As operators encounter increasingly demanding customer requirements and government regulations, developing effective Food Safety Management Systems (FSMSs) has become more important—and more difficult. Some manufacturers have encountered so-called data silos resulting from the use of multiple safety systems rather than a single solution. Breaking down these silos is key to managing data and processing steps across every aspect of an effective FSMS.
Written food safety management plans are an important part of that process, and manufacturers must have systems in place to enable rapid identification and tracking of each ingredient in all products through all processing steps. In the case of a recall or investigation, a processor should be able to show records of every step in an ingredient’s processing not only within the operation but one step forward and back as well.
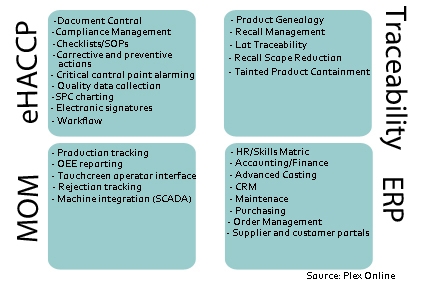
According to a recent white paper from Plex, the four key elements of an effective FSMS include HACCP plans, manufacturing operations management, instant traceability and recall management and single-source ERP.
To comply with FSMA regulations, producers must have documented HACCP plans as well as corrective actions/preventative actions available for inspection by FDA upon request, and maintain these records for at least two years. To comply with these regulations most efficiently, many manufacturers use FSMSs that support electronic document controls and signatures, including workflow management functions, checklists and standard operating procedures, and critical control point alarms. By automating data, this type of electronic FSMS enables compliance and saves cost while improving productivity.
Because the control of plant floor activities is a determinant of overall efficiency, more processors have started to use FSMSs that provide production tracking and scheduling, scaling and rejection tracking. Manufacturing operations management provides overall equipment effectiveness (OEE) reporting and analyses, helping producers avoid expensive analytics breakdowns while developing a technical knowledge base and responding quickly to maintenance problems. Best-in-class FSMSs go a step further by including human-machine interfaces and machine/equipment integration functionality that allow real-time visibility of inventory and processes.
The third major component of FSMSs is traceability and recall management. The rise in audits and recalls has reinforced the importance of one-up and one-back traceability, also referred to as an e-pedigree or product genealogy usage.
In the event of a quality problem, a distributor or customer can typically log in to its FSMS and create a report including the product’s unique identifying numbers. The report automatically includes an alert to the quality department, while also identifying the root cause of the incident and quarantining all that batch’s ingredients in the system. It also identifies shipped products needing to be recalled and sends notifications to all distributors and customers. Quick turnaround times comply with government regulations and allow processors to identify any ingredients early in the distribution chain, saving productivity and cost.
Finally, processors should engage enterprise resource planning (ERP) for a best-in-class FSMS. Traditionally, ERP has included only financing and accounting, but new ERP systems include advance costing, customer resource management, human resources, maintenance and purchasing. As ERP systems originated from more industrial manufacturing contexts, many are not well-suited to the food and beverage processing industry, making selection of the proper system extremely important.
Processors looking for complete FSMSs incorporating all four components may find single-source technology solutions. These solutions are often cloud-based, or software-as-a-service, systems that can enable cost savings through maintenance avoidance, easy data access and tracking and limitless storage. A complete FSMS solution can enable producers to focus on the next challenge on the horizon, rather than regulatory and quality enforcement.
To view and download a .pdf of the white paper, click here.




