Engineering R&D
Silver bullet slays bacteria, molds and fungi
Silver ions, impregnated throughout a substrate used in gaskets and seals, come to the surface to be exchanged with ions from microbes, rapidly and continuously inhibiting their growth.

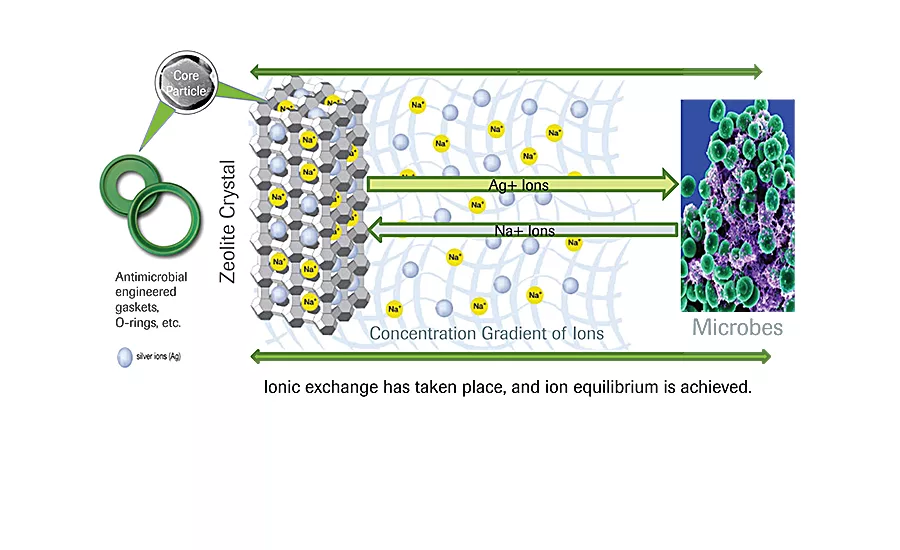
In the process environment, naturally occurring moisture on product surfaces activates a finely controlled release of Ag+ ions to exchange with other positive ions in the microbes. Source: Rubber Fab Antimicrobial Technologies.
Gaskets, seals and O-rings are used in the connection of pipes, inline flowmeters and machine inlets and outlets. Gaskets and seals prevent leaks in these static applications. But they may also provide an excellent edge (surface) to snag bacteria and help them grow, often creating a biofilm that can remain after inadequate or ineffective cleaning steps.
Silver has long been known to be an excellent weapon in killing bacteria and other microbes as its ions exchange with microbial ions, killing the microbes. The challenge, however, has been to find a better way than just coating a gasket or seal with a chemical or synthetic organic-based product that sloughs off during usage, contaminating product and leaving behind an unprotected seal surface. One solution to this problem is to permanently impregnate silver ions throughout the entire elastomeric matrix with a zeolite carrier, which allows silver ions to rise to the surface in a controlled release only when needed, preventing the growth of bacteria and other microbes.
Rubber Fab Antimicrobial Technologies has come up with proprietary formulated Nitrile/Buna, EPDM (ethylene propylene diene monomer) and silicone matrix materials that embed silver ions throughout. The materials, named ionAssure™, meet a host of regulations from EPA to FDA and achieve 4.5 log to 7 log reductions of Salmonella, Listeria and E. coli under industry standard protocol examinations. FE caught up with Bill Hurst, Rubber Fab president-Antimicrobial Technologies, to get a clearer description of this material and its applications. Hurst is a chemical engineer and business executive with experience in creating antimicrobial solutions for use in water filtration, food contact and medical device applications.
FE: Tell us a little about yourself.
Bill Hurst: My background is in commercializing technology-based and innovative solutions-oriented products in a broad range of markets including petro/specialty chemical, semiconductor, scientific instrument, food and water, and medical devices. I have been fortunate to be involved in the R&D and product development, sales and marketing, operations and general management of organizations from startups to Fortune 500 companies. Most recently, I have been focusing on developing new business and growth markets for antimicrobial-based solutions to combat dangerous and pathogenic microbes.
FE: How are ionAssure products made available for purchase?
Hurst: All ionAssure products sold will be 100 percent cured and ready for direct application. Gaskets, O-rings, cord stock and sheet material will be available for sale through our network of qualified distributors, consistent with current Rubber Fab product supply. This will ensure other critical performance requirements, such as dimensional tolerances, compression, tenacity, temperature/steam cycling ranges and durometers, are maintained or enhanced. It is also important to note that the ionAssure products will be interchangeable with our existing standard products.
FE: How does the ionAssure technology work?
Hurst: The key to ionAssure’s longevity and effectiveness is the infusion of silver ions in a specialized carrier particle. These multi-faceted zeolite carriers provide an efficient, three-dimensional release mechanism that delivers the ions independent of the particle’s orientation in the elastomer. Naturally occurring moisture from the process environment on the surface of the product activates a finely controlled release of Ag+ ions to exchange “on demand” with other positive ions [often sodium or -thiol groups] in the microbes. [See diagram.]
Once a localized equilibrium of ions within a few monolayers of water has populated the effective gradient zone, the ions are reserved until needed for ion exchange. Controlled ion release attacks the proteins/amino acids of the pathogen’s cell wall, allowing silver to invade the cell. The silver ions work together in a synergistic fashion, speeding cell wall penetration and maximizing effectiveness against the broadest range of microbes. As the ions bind to any microbes that may be present on the treated Rubber Fab products, they disable their critical functions, such as metabolism, respiration and reproduction.
FE: How does ionAssure differ from other available systems?
Hurst: This embedding of zeolite carriers—both at and below the surface—creates significant durability and safe process use versus topical alternatives. Topical chemicals, synthetic organics and other systems can easily degrade due to cleaning solutions or UV exposure, or be worn off with washings, abrasion, erosion or simple wear and tear. In contrast, each ionAssure product has a proprietary formulation employing continuous zeolite carriers throughout the products’ entire matrix. These noted stresses actually expose more zeolite carriers by design, improving the products antimicrobial action by accessing the additional reservoirs of Ag+, and increasing long-term performance and durability. As a point of emphasis, ionAssure’s durability and process safety also comes from our unique ability to release only the amount of ions that are required to maintain the antimicrobial effect. In contrast, other techniques may contaminate the process by sloughing off, continually off-gassing or leaching into the process environment regardless of need—thereby depleting their efficacy both from use, cleaning cycles and time, while also potentially imparting unwanted odor and flavor to customer products.
FE: How effective is the material in combating the growth of bacteria?
Hurst: Stringent testing (ASTM-2180 Direct Inoculation method) has proven ionAssure’s effectiveness, exceeding typical commercial requirements. For example, required third-party, antimicrobial performance testing showed a 7 log reduction of E. coli in Pt-cured silicone, a 6 log reduction of Salmonella in peroxide-cured Nitrile/Buna and a 6 log reduction in peroxide-cured EDPM elastomers. These reductions are at the surface of the elastomer; antimicrobial protection is limited to the treated surface and does not protect from a food product already contaminated with a pathogen.
FE: How well does the ionAssure material hold up to harsh CIP processes?
Hurst: Since the antimicrobial particles are embedded throughout the elastomer matrix, they provide a significant reservoir of antimicrobial action. Furthermore, the pore structure of the particles allows access to the entire reservoir of silver ions within the particle, regardless of what surface or corner is exposed to the microbial challenge. The end result is immediate, sustained and long-lasting action against microbes.
FE: What is the lifetime expectancy of a seal or O-ring made with ionAssure material?
Hurst: The antimicrobial protection is designed to last as long as the intended use-life of the gasket, O-ring or seal product. Standard operating practices for seal replacements will occur before the antimicrobial capabilities are exhausted.
FE: How can processors that do their own maintenance obtain these gaskets and O-rings?
Hurst: Rubber Fab sells through authorized distributors throughout the world. Processors and Original Equipment Manufacturers alike can be assured that our distributors will provide Rubber Fab products that meet all their requirements. If you have a specific application, please contact Rubber Fab, and we will direct you to a local distributor that will support your application needs.
For more information, www.rubberfab.com.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!






