Food Safety
Prerequisite programs ensure food safety
A regulatory shift that happened years ago remains a key part of food safety management today.
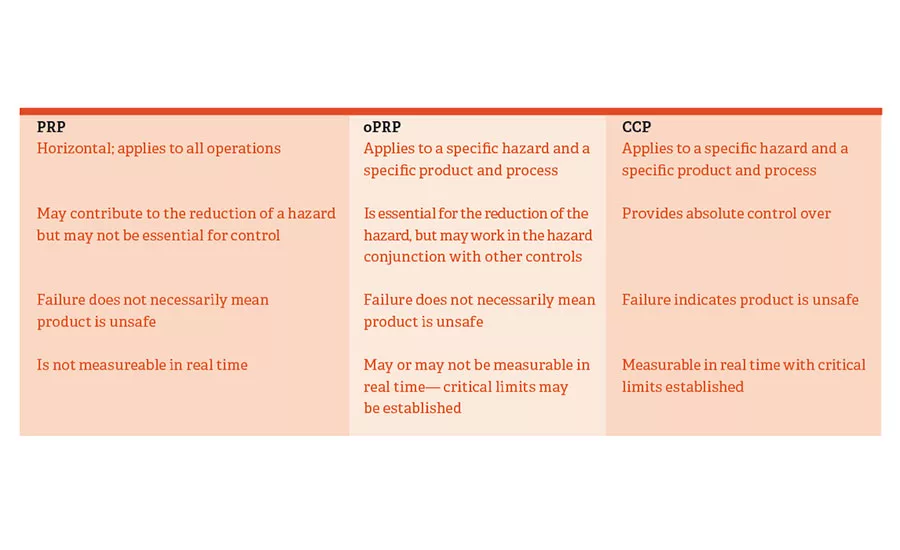
This chart is being used by ISO 22000 to clarify the difference between prerequisite programs, operational prerequisite programs and critical control points.
In 1995, the seafood HACCP regulation was enacted in the US and marked aArticles major milestone in the history of food safety.
First, it established that prerequisite programs could be used as a means to address potential food safety hazards. Persons working with HACCP programs in the ‘80s and early ‘90s undoubtedly remember plans that had 10, 15 or even 20 critical control points (CCPs). There were things in the plans that seemed essential for ensuring the production of safe foods: operations like product identification, cleaning and sanitizing, handwashing in ready-to-eat (RTE) operations and more.
These large plans were prone to failure and experienced regular deviations that, in reality, were probably not food safety failures. But remember, whenever there is a deviation at a point in the process that is deemed to be a CCP, the product is deemed potentially unsafe. In fact, the following statement from the preamble of the seafood HACCP regulation summarizes how this issue evolved:
“Sanitation, especially those issues related to cleaning and sanitizing and routine maintenance aimed at assuring safe operation of equipment, should now be included in the prerequisite program and not be considered to be CCPs.”
The regulation itself defined six categories of prerequisite programs (PRPs). These were preventive maintenance, product identification, product recall and traceability, establishment of Good Manufacturing Practices (GMP), development of the necessary sanitation standard operating procedures (SSOP) and education and training. The regulation also defined eight specific areas (which apply to all processors) where seafood processors needed to establish programs:
- Safety of water used in process or for manufacture of ice
- Condition and cleanliness of food contact surfaces, including utensils, gloves and garments
- Prevention of cross-contamination
- Maintenance of handwashing, hand sanitizing and toilet facilities
- Protection of food, food contact surfaces and packaging from adulteration
- Proper labeling storage and use of toxic compounds
- Control of employee health
- Exclusion of pests from the plant.
This enactment also made this approach to food safety go beyond just simply establishing CCPs. HACCP plans really evolved into food safety management systems (FSMSs), since all of these elements had to be integrated into them.
This system approach to food safety is part of all the Global Food Safety Initiative (GFSI) audit schemes, the ISO 22000 food safety standard, regulatory mandates for HACCP and FSMA.
So, even though utilizing prerequisite programs to ensure the production of safe food has been around for more than two decades, it is still something that creates confusion around the world. Prior to the release of the GFSI standards and the enactment of FSMA, the number one cause of product recall was the failure of PRPs. Even though food processors took care to meet the HACCP requirements, there were holes in their food safety systems when it came to PRPs.
ISO 22000 addressed this issue by ensuring sites verified the PRPs. In addition, prior to the release of the standard in September 2005, the committee debated not only how to address PRPs, but what to call them.
The standard specifically addressed this subject and established definitions for PRPs and for PRPs that one determines to be essential for food safety based on the hazard analysis. The latter were called Operational Prerequisite Programs or oPRPs. The definitions for the two types of prerequisite programs are:
PRP: Basic conditions and activities that are necessary to maintain a hygienic environment throughout the food chain suitable for the production, handling and provision of safe end products and safe food for human consumption.
Operational PRP: A PRP identified by the hazard analysis as essential in order to control the likelihood of introducing food safety hazards to and/or the contamination or proliferation of food safety hazards in the product(s) or in the processing environment.
This subject has been a bone of contention since the standard was issued in 2005, and the committee is working to clarify the distinction for potential users, in part by using the chart shown. The objective is to develop a standard that allows potential users to meet its requirements in a variety of ways depending upon their products, processes and facilities. There is simply no such thing as a “one-size-fits-all” standard in the food processing industry.
One possible clarification for this table might be to explain how each PRP, oPRP and CCP is measured or reported. A CCP generally has a value of some sort, such as time and temperature, flow rate, belt speed, etc. The PRPs or oPRPs often have a yes or no answer to them: Did you clean and sanitize? Is the proper label being applied to a product containing an allergen?
This clarification can even be applied to Preventive Controls (PCs) in the Preventive Controls Regulation (21 CFR Part 117). A CCP or a process PC has a value that must be validated according to the regulation.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!





