FSMA Violations 2020
FDA updates data on food facility inspection violations for FY2020
FSVP development tops the list with a 51% surge over FY2019 violations
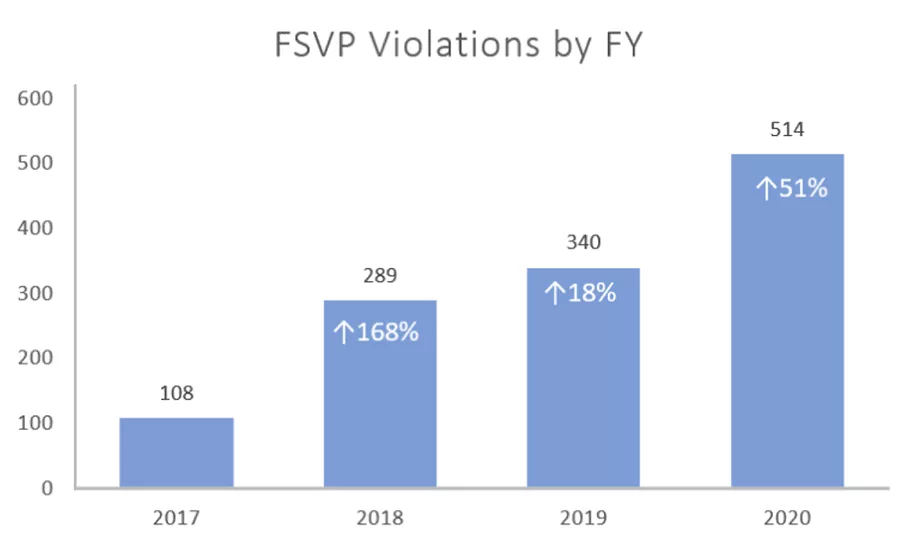
The number of foreign supplier verification program (FSVP) violations recorded since fiscal year 2017 (FY2017) has continued to increase, jumping 51% in 2020, compared to the previous FY2019. This graph was compiled by The Registrar Corp., based on FDA 2020 data.
The FDA released its annual Inspection Observation Data (raw data can be found here) for Fiscal Year (FY) 2020, and once again the data shows an increase year-by-year since 2017 of violations on the Foreign Supplier Verification Programs (FSVPs). The FDA began tracking FSVP violations in 2017, and the total number of violations in FY2017 was 108. In FY2020, the number had increased to 514 as reported by Registrar Corp, which has sorted out the data contained in the FDA’s Excel data file.
Under the FSMA, FDA requires most food importers to develop and maintain foreign supplier verification programs for their suppliers. The requirement helps ensure that suppliers remain compliant and are making products in a safe manner.
|
During FSVP inspections, FDA expects importers to present complete FSVPs that adequately assure suppliers’ food safety. In FY2020, FDA cited 514 facilities for failing to develop a FSVP. While FDA halted most of the food facility inspections for most of 2020 due to the COVID-19 pandemic, the agency continued to conduct FSVP inspections remotely, and FSVP citations increased 51% from 2019, when 340 violations were recorded. This is the third year in a row that failure to develop a FSVP was the top inspection violation, according to the Registrar findings.
Other violations loom
Number two on the list: In 2020, FDA cited 104 cases where facilities failed to provide hazard analysis, which includes potential biological, chemical or physical hazards that could occur at their facility as well as failed to establish a set of preventive controls for those hazards, according to the Registrar Corp.
Number three on Registrar’s compiled list of FDA violation data was the failure to meet compliance on preventing pests within a food facility—or misusing pesticides such that they could cause food contamination. These totaled 98 for FY2020.
Manufacturing, processing, packing, and holding controls account for 95 of 2020’s food facility citations—number four on the list. This citation indicates that a facility did not conduct operations under conditions that would minimize the chances for potential microorganism growth, allergen cross contamination, or contamination and deterioration of food.
The fifth most cited violation during food inspections in FY2020 were personnel issues. These can include failing to address hygiene issues or other good manufacturing practices in relation to employees handling food products. FDA issued 87 citations for this violation.
While no sanitation citations made it into the top five on their own, sanitation citations make up a significant portion of the FY 2020 violations when combined. For example:
- FDA cited seafood processing facilities 81 times for failure to properly monitor their sanitation practices. These include safety of water coming into contact with food, condition and cleanliness of food contact surfaces and/or maintenance of hand washing, hand sanitizing and toilet facilities.
- FDA cited 80 facilities for plant maintenance and sanitation issues. These are the result of failing to maintain a clean and sanitary food facility which can also pose environmental threats to food products.
- FDA cited facilities 45 times for sanitation of food contact services.
While FSVP violations saw a significant increase, the other top citations were issued fewer times in FY 2020 than in FY 2019. This is likely due to the temporary postponement of most food facility inspections in 2020 as a result of COVID 19.
For more information or help with complying with FDA regulations, contact Registrar Corp. at 757-224-0177 or visit www.registrarcorp.com/livehelp.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!







