Manufacturing News
NOSB votes on standards for next-generation GMO's
Next-generation genetic engineering techniques include the use of synthetic biology.
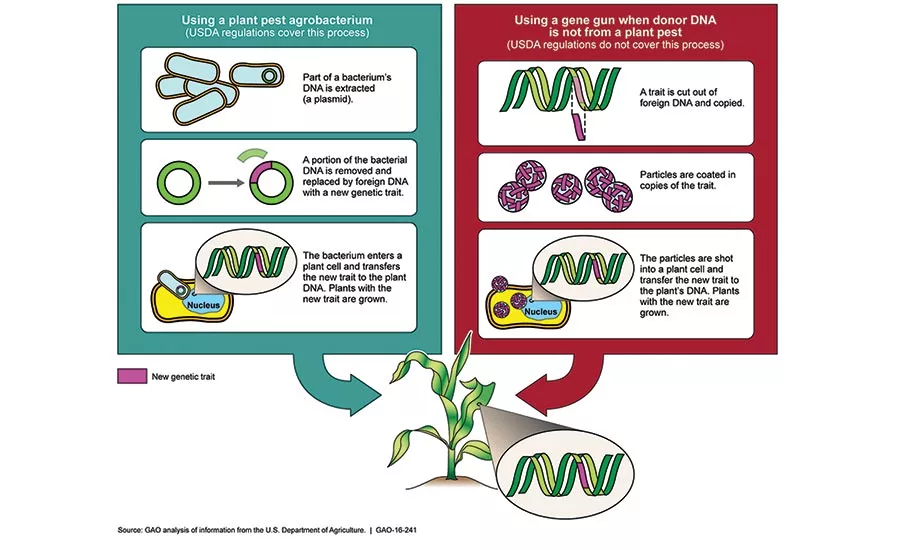
This diagram shows an example of a genetic engineering technology with and without the use of a plant pest and the extent of the USDA regulations. Source: GAO.
With “first-generation” GMOs in practically 90 percent of the crops we grow, the National Organic Standards Board (NOSB) voted unanimously to recommend an update to US organic standards to exclude ingredients derived from “next-generation” genetic engineering and gene editing. Next-generation genetic engineering techniques include the use of synthetic biology, which is a new set of genetic manipulation methodologies that include using synthetic DNA to re-engineer organisms to produce substances they would not normally make or editing DNA so as to silence the expression of certain traits, according to the Center for Food Safety.
This recommendation to the USDA’s National Organic Program will ensure that ingredients derived from these new genetic engineering techniques will not be allowed in the production or final product of foods and beverages that are certified organic, according to Dana Peris, food and technology policy campaigner with Friends of the Earth (FOE).
“The Board’s hard-fought proactive stance on synthetic biology will both help preserve the integrity of organic standards and raise awareness about this virtually unregulated and unlabeled form of genetic engineering,” Peris says. “It’s critical that organic standards treat new types of genetic engineering that are rapidly entering our food and consumer products as rigorously as the first generation of GMOs.”
The consumer groups believe that, like traditional GMOs, synthetic biology ingredients are entering food and consumer products in the absence of adequate health and environmental safety assessments, oversight and labeling. Many of the new second-generation GMOs are also being falsely marketed as “natural.” For example, some of these synthetically developed products include stevia, coconut and cacao, which are meant to replace plant-based ingredients—many of which are currently produced by small farmers in the Southern Hemisphere, whose livelihoods may be displaced by synthetic biology crops. FOE lists other synthetically genetically engineered (GE) products to include gene-silenced apples, CRISPR waxy corn and Cibus Canola oil.
“The National Organics Standards Board has made clear that all kinds of genetic engineering are to be excluded from ‘organic,’” says Jaydee Hanson, senior policy analyst at Center for Food Safety. “The public expects the government to actually assess the new foods that it is permitting on the market. Unfortunately, the government has failed to update its regulations to adequately assess these new kinds of genetic engineering. When the USDA approves that NOSB recommendation, consumers who want to avoid GMOs will be able to use the Organic Seal to know that the product is not a GMO.”
Irrespective as to whether this new generation of GMOs appears to affect human health negatively, there is still a practical problem with these products mixing with non-GMO varieties, and USDA finds itself with a situation that neither EPA nor FDA have in regulating these products. Both EPA and FDA regulate these products based on potential negative affects to humans and/or animals.
USDA has no regulations for these new genetic products because they don’t resemble the more traditional first-generation GMO products, according to the Government Accountability Office (GAO). Under current regulations, USDA restricts the introduction and dissemination of GE crops for which the donor, vector or recipient of genetic material is a plant pest—such as a bacterium or virus—until the agency assesses certain potential plant and environmental health risks and determines the regulated article does not pose a potential pest risk.
For more information:
“Genetically Engineered Crops: USDA Needs to Enhance
Oversight and Better Understand Impacts of Unintended Mixing with Other Crops,”
GAO Report GAO-16-241, 2016; www.gao.gov/assets/680/675791.pdf.
“Exogenous plant MIR168a specifically targets mammalian
LDLRAP1: evidence of cross-kingdom regulation by microRNA,”
“Cell Research” (2012) 22:107–126. doi:10.1038/cr.2011.158; published online September 20, 2011.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!







