Food Safety
Food Safety at the Crossroads
The recent peanut paste recall-largest ever in terms of supply chain-points to the urgency of food safety today

Peanut butter and several products made with peanut paste could have had salmonella due to improper handling of peanut paste at the Peanut Corporation of America. Image by Shutterbug75 from Pixabay
.
It’s 2:00 pm, Friday, January 23, 2009, when the phone rings. I see it’s an 800 number, and I pick it up, expecting some product solicitation. Instead, it’s Costco warning me that the carton of Clif bars my wife purchased two months ago had been recalled due to possible Salmonella typhimurium contamination. I already knew Clif bars had been recalled because I received the FDA recall list the day before, but I had forgotten completely about my wife’s purchase.
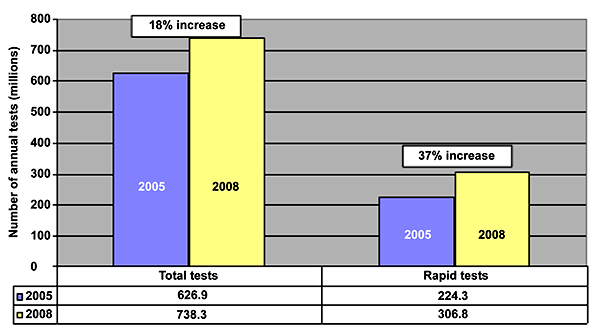
My experience exemplifies what one part of the food safety supply chain is about: not only knowing where contaminated food originated, but also protecting those at the “fork” end of the “farm-to-fork” cycle. For Costco’s Assistant Vice President of Food Safety Craig Wilson, this quick response was easy to implement. When I spoke to him a few days later, he told me that as soon as he got word from the supplier, he was able to pull the list of Costco members that had purchased Clif bars between July 1, 2008, and January 20, 2009, and send it to his third-party affiliate to make the automated calls. Costco isn’t the only retailer that handles recalls this way-Wegmans warned its Shoppers Club card members of recalled products in the same fashion.
Back to basics
Despite the sophisticated lab detection equipment available to check for pathogens, adulterants and other chemicals, this event-like the Chinese melamine contamination- never should have happened. Certainly, Peanut Corporation of America (PCA) should have learned its lesson from the earlier ConAgra/Peter Pan Salmonella incident, says Center for Food Safety Policy Analyst Jaydee Hanson. There is only one way that peanuts can become infected with Salmonella, he adds, and that’s through sick animals that are Salmonella carriers-namely birds and rodents.
According to a January 27, 2009, FDA “483” form (FEI #1036857), posted on its Web site, several FDA-conducted observations at PCA’s Blakely, GA, plant pointed to known positive tests for Salmonella, but PCA still shipped product from late September 2008 into January 2009. In addition, the document found “failure to maintain equipment, containers and utensils used to convey, hold and store food in a manner that protects against contamination.” Further observations revealed an ineffective roasting stage (a critical control point (CCP) to kill pathogens), mold growth on ceilings and walls, gaps in A/C intakes on the roof where water leaked in over production and packaging areas and other exposed openings to the outdoors.
While testing revealed the presence of Salmonella typhimurium and several other serotypes, quality can not be increased through testing, says William Pape, executive vice president and founder of TraceGains, a traceability software company. “One of the most important things we need to do is to change our paradigm that we can test safety and security into our food system,” he adds. Processors must design in safe processes and then monitor them for continuous compliance. Pape contends that over-regulation won’t necessarily help either, because processors can become lax about quality, thinking food safety is the regulators’ job. Rather, he says, streamlining the regulatory infrastructure makes good sense as long as there is a modicum of oversight in food processing operations, and a government regulatory database that could be used to perform trace-back and trace-forward analyses.
“Lab sampling plans and audits statistically will not catch all the issues,” says Carl Deely, managing director of TBM Australia. “Problems will and do get to the consumer, therefore, prevention must start at the source, not just with the raw material supplier or farmer, but at each transformation step in the process and with each employee.”
The problem with microbiological testing is results take too long, says Tom Weschler, principal of Strategic Consulting. Processors know that by employing HACCP principles: control the raw material and control the process, then the product will also be under control. But it’s difficult to hold up production while waiting for test results on incoming ingredients.
“HACCP is effective only when a food processing plant has effective prerequisite systems,” says John Surak, Ph.D., principal of Surak and Associates. “The FDA’s 483 shows numerous failures in the plant to meet the basic good manufacturing practice (GMP) requirements.”
“We need more process control and we have to regulate on process control,” says Surak. “That’s not to say we should stop other regulations,” he adds. ISO 22000 provides a sound basis for linking together food safety management, ISO 9001 and HACCP principles. Most important, says Surak, is to make food safety a strategic issue for senior level management. And while process control is important, “technology won’t save us,” he says. “Food safety has to be built into the process of food from farm to fork.”
Facing the enemy
While melamine and Salmonella have taken center stage for the last year, there are plenty of other nefarious contaminants waiting in the wings. For analytical equipment suppliers and solution providers, developing a marketing strategy based on current trends helps in predicting possible future issues. Gerry Broski, director of food safety marketing at Thermo Fisher Scientific, looks at contamination first from how it’s acquired in the production process. For example, he’s broken it down into the following classes:
- Accidental (could be chemicals or bacteria)
- Deliberate (such as melamine)
- Inherent (such as mycotoxins from molds and fungi)
- Part of the process (such as acrylamide)
- Environmental (such as water dripping into a production line).
Besides melamine, plenty of other chemicals contaminate food and beverage products, ranging from heavy metals in the ground and oceans, to mycotoxins, residual pesticides and human antibiotics.
Sometimes testing reveals unexpected results. Jerry Zweigenbaum, Ph.D., food industry applications/solutions development manager at Agilent, points to a study entitled Determination of Pesticide Residues in Fruit-Based Soft Drinks, published in the December 15 issue of ACS’ Analytical Chemistry journal.1 The study, which used liquid chromatography-electrospray time-of-flight mass spectrometry (LC-TOF MS), found fruit-based soft drinks in Spain and the UK contain the highest level of pesticides, and samples from the US and Russia were among the lowest. The reason is ironic. In Europe, unlike the US and Russia, natural flavors are preferred to the use of artificial flavors. Natural flavors, e.g., orange, are obtained from the orange rind, which is usually treated with fungicides to prevent mold that could generate mycotoxins if it grows on the rind. While the fungicide doesn’t penetrate the skin to the actual fruit, the residual level of fungicide on the orange rind will be multiplied as the rinds of several oranges are used in making a natural flavor, causing the abnormally high concentration in the soda.
Zweigenbaum has run his own tests on oranges from the local grocery and found them to have thiabendazole (a fungicide) on the skins, but the level was within the maximum residue level (MRL) set by the FDA. Why the concern with mycotoxins? The aflatoxins found in corn fungi are harmful or fatal to livestock and carcinogenic to humans. Levels higher than 20 parts per billion (ppb) are considered unsafe by FDA, and one contaminated grain in a five-pound sample can result in levels of 20 ppb or more. Patulin (a mycotoxin created by mold growth on apples) levels in apple juice are set by FDA at a maximum of 50 ppb in 100% apple juice and are measured using a gas chromatograph/mass spectrometer (GC/MS).
Analyze this
While the FDA requires traditional, time consuming culture tests for bacteria, processors would like to find faster ways of bacteria screening, especially in incoming materials, says Weschler. If processors could get faster results from these indicator tests, then they would have a better sense of their process and how to control it. “Food companies produce at risk. They have no idea what’s in their raw materials. All they know is they have a certificate of analysis (COA) from the company that produced the starter materials,” adds Weschler.
According to Mary McBride, Ph.D., manager, molecular preparation, Agilent Laboratories, there are automated testing technologies that allow rapid genetic analysis, such as polymerase chain reaction (PCR), which is fully capable of screening for E. coli O157:H7 as well as Shiga toxin-producing Escherichia coli (STEC) and enteropathogenic E. coli (EPEC). However, FDA’s protocol mandates a “culture confirmatory.” That is, says McBride, you have to actually grow the bug and confirm it via culture. It’s not a true positive until the culture shows that bacteria are viable.
Yolanda Fintschenko Ph.D., Thermo Fisher manager, food safety technologies group, suggests that culturing still is a good idea. Molecular techniques such as immunoassay and genetic techniques like PCR give very precise species identification. But there are two things these tests can’t reveal. First, the tests can’t determine whether the samples are viable. Will they reproduce? For example, disinfected material may contain Salmonella, but it’s not growing. When trying to trace the origin of a contamination, it’s really important to know the origin of the growing pathogen sample. Otherwise an investigator could be following false leads.
The other point Fintschenko makes is biochemical testing has the ability to determine what serotypes of the bacteria may be growing. Typically you can’t get those results without biochemical testing. Scientists at FDA, CDC and the Agricultural Research Service (ARS) are developing instrumental techniques to get to that answer faster by looking at indicators such as metabolites and proteins that can be used to rapidly screen the serotypes. But now the gold standard is the biochemical test to determine the serotype, says Fintschenko. And it’s the serotype that’s key to tracing, for example, which Salmonella is involved in a recall.
Speed tracer
While speeding up tests and results can’t improve quality, it potentially can speed up production. Weschler says there are three parts to any microbiological test: sample preparation, detection and reporting the information. In the past 20 years, test suppliers have concentrated on speeding up the detection stage-whether it’s traditional testing, antibody-based (immunoassay) or more recently the molecular methods. But over the last couple of years, the focus has been on the sample prep stage-how to have better enrichment to grow the bacteria faster.
Broski says 60% of analysis time is spent in sample preparation and his company is analyzing work flows to find ways to shorten the sample prep time. One method in enzyme-linked immuno-sorbent assay (ELISA) screens was to delete an extraction method, and by using centrifugation, still come up with excellent sensitivity and increased throughput. Fintschenko pointed to turbulent flow chromatography as an automated sample prep technique that selectively removes small molecules in a matrix of large molecules as one technology that speed analysis.
According to George Tice, DuPont Qualicon director of R&D, DuPont’s BAX system assays include Salmonella, Listeria species, L. monocytogenes, E. sakazakii, Campylobacter, Staph aureus and yeast and mold. Most take one to 3.5 hours and are used on samples that have been enriched for eight to 48 hours. “However, our reverse-transcriptase PCR test for Listeria on environmental surfaces provides results within eight hours of sample collection, and this year we expect to introduce a new test for Vibrio in seafood that gives results in less than 24 hours,” Tice says. The goal is to reduce system time so tests can produce a result by the end of a shift. Some ways of doing this would be to reduce the cycling time to less than one hour and increase the assay sensitivity by concentrating the sample.
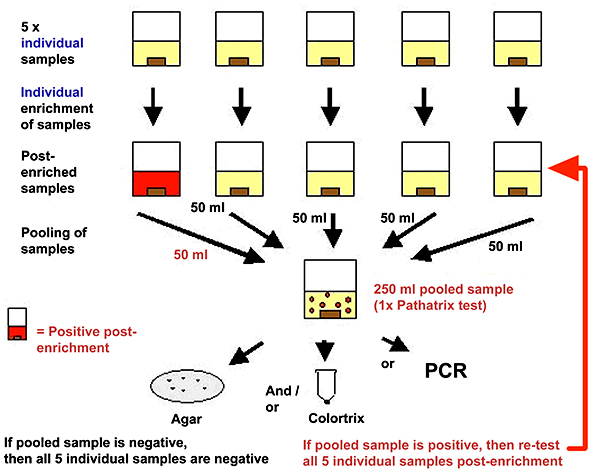
Cadbury Schweppes plc adopted the Pathatrix pooling system from Matrix MicroScience for its routine pathogen testing and has achieved increased sample throughput and more timely results. The system analyzes the entire, standard-sized 225 ml + 25 g sample simultaneously by re-circulating the sample through a capture phase where the antibody-coated magnetic beads are immobilized. According to Jeff Banks, Ph.D, group director of food safety and quality for Cadbury Schweppes, “The system provides a validated, science-based solution to the rigorous demands of a highly independent and time-critical supply chain. The system integrates well with other technologies and provides a high quality and practical asset for our laboratories.”
Melamine, chemicals and metals
In 1958, the US Patent Office granted a patent (2,819,968) entitled “Feed Composition” to The Dow Chemical Co. in Midland, MI. The patent presented melamine use in various cattle feed mixtures as a non-protein nitrogen (NPN) source. Cows’ ruminant digestive systems may be able to process melamine safely, unlike humans and pets for which it is a poison, says Surak. Further research at the University of Georgia, College of Agriculture in 1978 suggested that melamine might not be an acceptable NPN source for ruminants because it wasn’t as effective as other materials in converting to ruminal protein.2
While melamine was added to gluten and Chinese dairy products to fake increased protein levels, finding melamine for the first time presented challenges. Because protein levels (which are based on nitrogen) in food typically are checked using a simple and relatively inexpensive total Kjehldahl nitrogen (TKN) test, an NPN such as melamine shows an increased level of protein when added to food. By its nature, a TKN test assumes there is very little NPN present. When using analytical equipment, the problem in finding melamine begins with extraction, says Zweigenbaum. Since melamine is not very soluble, if you were to attempt extraction with methanol, for example, you wouldn’t extract any melamine so the test would appear to be negative.
Fintschenko advises using FDA’s recommendation to make melamine determinations: a liquid chromatography triple quadrupole tandem mass spectrometer (LC-MS/MS). (See FDA Laboratory Information Bulletins 4421, 4422 and 4423.) This method, says Fintschenko, is quite selective for melamine and its metabolites as well as cyanuric acid.
Finding the unknown is always the challenge, and discovering residual levels of heavy metals would be difficult or impossible with GC/MS or LC/MS technology, yet this technology could screen for any of the 800 to 850 pesticides used in the world, says Zweigenbaum. For heavy metals, elemental analysis with an inductively-coupled plasma mass spectrometer is the instrumentation of choice, according to Fintschenko. This technology has the ability to detect levels in the parts per trillion (ppt) level.
Data communication
While there has been a real urgency in getting faster results from tested samples, sometimes there’s a communication problem between the laboratory information system (LIMS) and the plant manufacturing execution system (MES) and/or the ERP system, says Broski. “As a major manufacturer of LIMS, we understand the criticality of tracking the sample as it goes through the laboratory analysis process. The output of the analysis must be actionable and link back to the plant manufacturing system. If data winds up in a report that takes 15 days to process, it will be of no immediate use.” Processors with well-connected systems have a better chance of saving precious time and isolating a product if there is an indication of contamination.
According to Wonderware MES/EMI Marketing Programs Manager Maryanne Steidinger, LIMS is the system of record that captures all of the quality tests and results that are needed before a product advances to the next line or packaging, at which time MES becomes the system of record. While this data is good for improving yield, some data from LIMS would connect to MES for track and trace.
Track and trace will mature to a level that industry standards like the Produce Traceability Initiative (PTI) and GS1 will allow consumers the ability to learn about the origins of the components in a bag of mixed greens, says Kerry Farrell, VP of sales and marketing at FoodLogiQ. In the future, consumers will be able to scan codes and learn more information about a product, and even opt for cell phone notices that link back to product manufacturers.
Seneca Foods Inc. has seen customer information requests and plant audits grow from 193 in 2003 to 2,835 last year. Seneca partnered with FoodLogiQ to develop an agricultural information management system (AIMS) that will provide trace back from the can to the field. The system supports more than 4,000 fields, 2,500 growers and more than 500,000 acres in 12 states.
FDA or Wal-Mart?
After the PCA Salmonella fiasco, many people, including President Obama, have ideas about what is wrong with FDA and how to fix it. But, it won’t be FDA that makes or breaks a food processor. “The influence on food processors is not just FDA or USDA,” says Weschler. “It’s really the influence of the food retailers and the service companies, and they will dictate what test methods will be done for them to feel comfortable that they can sell your product safely.”
Farrell describes how her company is helping processors meet the Global Food Safety Initiative (GFSI) and the Wal-Mart mandate, which was released on February 4, 2009, to require suppliers of its private label and other food products to have their plants certified against one of the internationally-recognized GFSI standards.
According to Andrea Thomas, Wal-Mart’s senior vice president, private brands, “The GFSI standards are an added step that will help us-and our US food producers-keep our quality commitment.”
J.P. Suarez, Wal-Mart’s senior vice president and chief compliance officer adds, “The requirement for suppliers to complete these certifications demonstrates our leadership in food safety and our commitment to global safety standards.”
Whether or not you supply to Wal-Mart, you determine the future of your company by the food safety practices you follow. You can spend hundreds of thousands of dollars for state-of-the-art analytical equipment and/or testing, but following good manufacturing and/or agricultural practices will bring the test results you want to see-a safe food product.
For more information:
Tom Weschler, Strategic Consulting, 802-457-9933, weschler@strategoc-consult.com
William Pape, TraceGains, 303-884-5693, will.pape@tracegains.com
John Surak, Surak and Associates, 864-506-2190, jgsurak@yahoo.com
Gerry Broski, Thermo Fisher Scientific, 763-783-2600, gerry.broski@thermofisher.com
Carl Deely, TBM Australia, 800-438-5535, cdeely@tbmcg.com
Jerry Zweigenbaum, Agilent, 800-227-9770, j_zweigenbaum@agilent.com
Mary McBride, Agilent, 408-553-7473, mary_mcbride@agilent.com
Yolanda Fintschenko, Thermo Fisher Scientific, 408-307-1328, yolanda.fintschenko@thermofisher.com
George Tice, DuPont Qualicon, 302-695-5300
Maryanne Steidinger, Wonderware, 949-639-8713, maryanne.steidinger@wonderware.com
Kerry Farrell, FoodLogiQ, 416-452-8742, kfarrell@foodlogiq.com
Mark Offerhaus, EBI Food Safety, +31 654-913-176, m.offerhaus@ebifoodsafety.com
References:
1. Juan F. Garcia-Reyes, Bienvenida Gilbert-Lopez and Antonio Molina-Diaz, “Determination of Pesticide Residues in Fruit-Based Soft Drinks,” Analytical Chemistry, 2008, 80 (23), pp 8966–8974.
2. G. L. Newton and P. R. Utley, “Melamine as a Dietary Nitrogen Source for Ruminants,” Journal of Animal Science, Vol. 47, No. 6 (1978).
New ways to fight pathogens
According to Nandini Ishwarakrishnan, industry analyst at Frost & Sullivan, some new technologies combined with older applications will help fight pathogens in the future. Nanoparticles, micro and/or nano encapsulation techniques and the development of antimicrobial edible films, silver nanoparticles and bacteriophages represent both already realized products and others under development.
It is well known that silver kills bacteria, says Ishwarakrishnan. Silver nanoparticles usually are made by vaporizing silver in the presence of an inert gas, and later compressing/condensing them under controlled conditions to the size of nanoparticles. Silver nanoparticles effectively cut off the oxygen supply of bacteria, and while research has found them to be effective in coatings and packaging material, it’s too early to tell whether they’d be safe to ingest.
Mark Offerhaus, CEO of EBI Food Safety (Netherlands), has some reservations. It is suggested that incorporating silver nanoparticles in materials will immobilize the silver and still generate the beneficial antimicrobial effect. This doesn’t mean that all the dangers for the environment and human health are eliminated, but the technology definitely warrants more research, Offerhaus says.
He expects that phage-based pathogen detection methods are likely to be used more extensively in the food industry. Several different detection strategies exist, but all take advantage of inherent phage properties: host specificity, specificity for living target organisms and ability to replicate faster and at higher numbers than their host cell.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!






