Food Safety
Using software to track contamination
Software tools provide more accurate tracking, tracing and elimination of contamination.
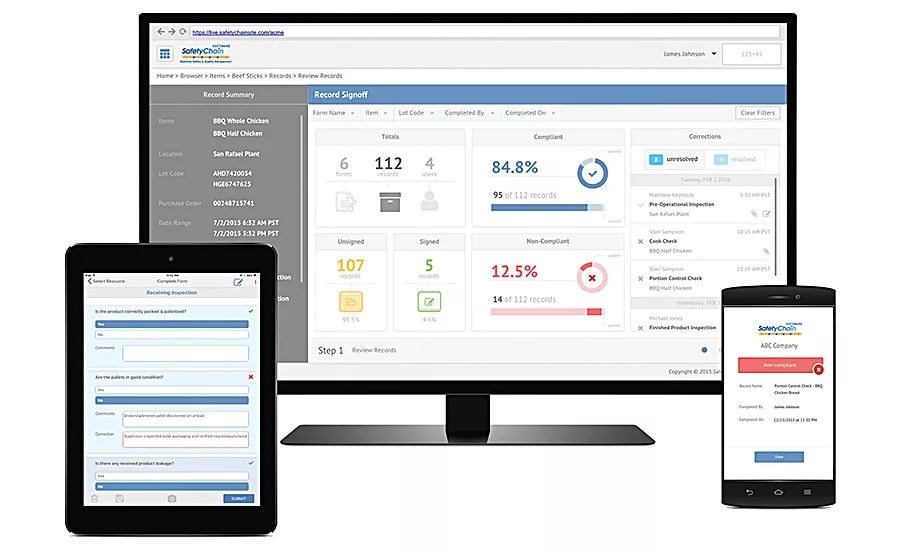
SafetyChain’s solutions deliver program visibility, data intelligence and critical tools to more effectively manage food safety and quality. Source: SafetyChain.

Built on the SAP ERP platform and using SAP’s OpenText technologies, Deloitte’s Food Quality and Safety Solution features digital records management, food safety audits management, enhanced traceability and recall and CAPA. The components integrate seamlessly with one another, providing a cohesive system for managing quality, safety and compliance in operations. Source: Deloitte Consulting LLP.

SYSPRO’s ERP is available on any hardware platform, making it easy for everyone in the enterprise to have process data when and where it’s needed. Source: SYSPRO USA.

It is important to look at food safety, quality and efficiency metrics in concert. Typically, these are siloed categories. Having a holistic view of metrics can ensure that one area of focus does not compromise the other. Sage Clarity finds that when software systems come together through integration tactics with one view, improvement objectives are accelerated. Source: Sage Clarity.
With the food supply chain becoming increasingly global and adding extra complexities to manufacturing, more accurate food safety tools need to be employed to keep bad products from reaching other food processors as ingredients or consumers as end products. While food and beverage processors need to deliver food-safe products to their customers, they’re also feeling the need to get these products out the door more efficiently while maintaining high quality.
These needs don’t have to work against each other. But, if you’re still using antiquated manual methods to keep track of your suppliers and ingredients plus your customers and distributors while trying to monitor quality and produce a safe product, it’s time to automate—before you lose your mind and potentially your business to a recall caused by an “accident” or “problem” you overlooked. Software tools can efficiently help manage documents and records to meet FSMA/USDA demands, monitor traceability and recalls, provide corrective and preventive action (CAPA) and handle food safety audit management, ensuring high-quality standards.
Do some research!
Software solutions exist for just about any size company and application(s). First, know your operation thoroughly and give some thought to growth. Second, keep it simple if you’re just getting started in automation. Automate in a way that fits your business, understanding and future.
For larger companies with ERP and/or MES systems installed, much the same is true when adding capabilities—because you don’t want to duplicate functionality or leave any gaping holes where you need capabilities the most. In most all cases, automation suppliers and/or system integrators can help.
“For most small processors, a clipboard and paper recordkeeping is the standard,” says Maryanne Steidinger, Schneider Electric director, industry solutions marketing. “These manufacturers traditionally use manually driven processes, so they would need to look at systems that could gradually replace their paper processes without a huge impact on the organization or a requirement for new staffing and skills.”
Certainly, some training will be required with any new technology purchase, but it should be gradual, and it should be thoughtful in its implementation. For automation beginners, Steidinger says Schneider Electric would normally recommend a data historian and low-end HMI. The HMI would provide process visualization, and the historian would give a processor an electronic system of record to use for reporting and some rudimentary analysis.
“No matter the size of manufacturer, it does not make sense anymore with today’s regulatory pressure to maintain product quality history on paper,” says Jon Simpson, Sage Clarity director, advisory services. “The cost of doing business now requires immediate access to event records as product moves throughout the supply chain…We find small and mid-sized processors must have strong management principles using continuous improvement and shop floor readiness to embrace change.”
Moving toward electronic
“It’s important for companies to decide first what are the objectives of leveraging technology and getting rid of paper in the first place,” says Dan Bernkopf, SafetyChain Software VP FSQA solutions. “Is it to ensure programs are being properly documented for audits/compliance? Is it to improve FSQA [food safety/quality assurance] performance and mitigate risks? Save costs in overseeing program execution?”
Processors need to prioritize those objectives when shopping for a vendor. Also, companies need to take into consideration the time and resources it takes to properly set up a system both internally and externally. Regardless of the size of processors, and whether they’re growers, processors, manufacturers or retailers, all have FSQA protocols—internal (customers) and external requirements (FSMA, USDA, GFSI, etc.)—to which they must adhere and properly document with the appropriate records, adds Bernkopf.
Jack Payne, vice president, product management and solutions consulting at Aptean, suggests that processors follow three basic steps in moving from paper recordkeeping to software systems, beginning with assessing their preventive processes. This means initiating a full process audit, analyzing what’s working and not working with regard to food preparation, packaging and transportation. This analysis can help a processor gain greater visibility and heighten awareness of vulnerable areas before they become hazardous. By identifying the risk areas within the manufacturing process, an organization can work to correct them and realistically establish a plan that’s better suited for prevention and fast action.
Jason Dea, director of product marketing at Intelex, suggests including people to this first auditing step because people are part of the process and the ones responsible for it.
“By their very nature, software systems, including those designed to help manage compliance, are there to automate and accelerate existing business processes,” he says. “If those processes are not well defined or inconsistent, this can introduce significant risk to an organization. Even worse, what some organizations discover is that if those business processes are flawed while ‘on paper,’ software will, in fact, accelerate and expose those flaws and inconsistencies further.”
“The second step,” Payne says, “is to implement technology like an ERP system with full backward and forward lot trace/recall capabilities. Finally, certification through programs like the Global Food Safety Initiative [GFSI] and Safe Quality Food [SQF] can also better prime an organization for initial and ongoing FSMA compliance.” Also, GFSI certifications—though not mandated by law—were designed with the same principles as FSMA, making them terrific guideposts for processors to use as a way of self-policing compliance.
“The requirements of FSMA have caused all companies to reevaluate their approach to food safety and quality management,” says Mike Edgett, Infor industry & solution strategy director.
The first step for many smaller companies to move beyond paper is focused on managing the quality records of incoming raw materials and finished goods. They move to a quality management system (QMS) that can store the analysis of incoming ingredients (both those provided by the supplier as well as testing done in house) and testing done post production. This provides a system to easily pull records as required, but the underlying driver isn’t just electronic records. It’s also the stop-gap measures and formalized processes that an integrated QMS can provide.
“It will ensure that a product is not accepted or released if it falls outside of the accepted range,” adds Edgett.
“Product specifications used to be stored as documents tailored for human consumption,” says Dag Heggelund, Trace Register executive vice president and chief technology officer. “Each member of the supply chain is expected to adhere to both their own specifications and the specifications of the buyer. Since specifications are used by supply chain members to differentiate themselves, large variations in specifications exist in the food industry… By digitizing the specifications and product information, it lets computer programs process the data. This enables both sellers and buyers to have increased confidence that their products meet all of their specifications all of the time.”
SYSPRO USA assists processors in transitioning from manually administered paper processes into an electronic software system through an implementation plan that places the major emphasis on defining and documenting a company’s business flows and quality processes.
“Once defined, these business flows and quality procedures are documented and presented [to the processor] for approval,” says Joey Benadretti, SYSPRO USA president.
Upon approval, a business model incorporating the processor’s business flows and quality procedures is built to provide an effective proof of concept. Once the model has been approved, training on all procedures begins, and when completed, a stress test evaluation of the company’s personnel ensures all employees understand how to use the software. The implementation typically ends with a go-live and monitoring of the system for a month by the software vendor to ensure the system is working properly. With each implementation, a professional business consultant and a project manager are assigned to keep the system on track and to consult with the processor.
Speaking about quality
“Food quality starts with quality ingredients,” says Scott Deakins, Deacom Inc. COO. “Quality ingredients are derived from quality vendors. We find that manufacturers that consistently provide quality control feedback to their vendors are able to drive the best results. To do this, implement vendor scorecards, which rate vendors based on the quality, timeliness and accuracy of their shipments.”
If a vendor scorecard is not an option, then a processor’s purchasing team should take the time to provide direct feedback via phone or email.
“Quality is simply a measure of how well a product meets a set of specifications,” says Heggelund. Spot checking incoming ingredients works well for large volumes, but changes in inventory management policies are resulting in more frequent and much smaller shipments, which reduce its effectiveness. “Automated quality assurance software is needed to ensure that every product meets all the requirements of both the buyer and the seller.”
Problem is: ingredient quality is very supply chain dependent.
“These days, food processors’ supply chains are more complex. Raw ingredients can be sourced from all over the country, if not the world,” says Intelex’s Dea.
As a processor scales its production, it becomes increasingly challenging to manage all of the information related to what comes from where and what’s going on where in a simple spreadsheet or homegrown database. While these systems might work initially, the maintenance and support of them can be a significant roadblock to success.
“These days, there are not only off-the-shelf tools for supplier relationship management or product specifications management, but many of these tools are also cloud based,” adds Dea. These systems allow processors to get a system up and running—and seeing value—more quickly than in the past with building on-premise systems.
Cloud or software-as-a-service (SaaS) solutions are becoming more prevalent. As small processors may not have the capital to invest in new IT infrastructure that may be required for adopting some quality management software systems, there are solutions that can be hosted in the cloud for access from any device, at any time, says Sage Clarity’s Simpson. In addition, besides providing an alarm when product is out of spec, QMS software can monitor product characteristics, providing a means of control.
“The earlier a manufacturer can capture nonconformance, the better chance they’ll have of not having critical quality failures,” says Schneider’s Steidinger. “Quality does start with raw materials—incoming inspection can ensure ingredients meet specifications, and a system of record such as a MES or batch system can capture time/date and store incoming inspection data for reference.”
In terms of cost, Steidinger points to newer, lighter-weight batch products (e.g., Wonderware Recipe Manager Plus) that take much less IT time and configuration and are suited for a foundational operations management system. HMIs can also be pressed into duty to a certain degree to view process alarms and foretell quality issues.
Sensible software acquisitions
Processors often buy software over time—and accumulate redundancies—and still have holes in coverage plus hardware that’s not tied into any software. Are there any solutions to this dilemma?
“It’s true—unless we are talking about a new startup, it’s likely that a company has a collection of systems—some software based and some not—that try their best to work together,” says Khris Kammer, Rockwell Automation information partner and competency manager. “The inevitable overlap is only mitigated by how each company uses certain software functionalities.”
“We think a software strategy of starting small, and incrementally adding features as you grow, is the best way to manage a robust IT infrastructure that meets the needs of the organization at that specific time in their maturity,” says Steidinger. “To buy a set of products without the internal readiness [training, funding] will ensure a failed implementation.”
“Key here is redundancy,” says SafetyChain’s Bernkopf. “With point solutions, redundancy can be one of the hardest hurdles to manage, especially if the solutions don’t talk to each other, share data and deliver the data in a meaningful way for FSQA teams to act upon effectively and efficiently.”
“The ability to exchange data between different applications is essential,” says Heggelund. A key challenge in application data exchange is “entity resolution” or “record linkage.” Product item codes may differ between departments and between applications. In addition, different departments may have unique terminologies and ontologies. Exact transform and load (ETL) software programs are specifically designed to make this process easier.
“The issue with a patchwork of solutions, some of which meet your needs and others with holes, is the reason we are seeing more companies moving to a cloud-based ERP solution with optional modules,” says Infor’s Edgett. “This enables them to make sure they are always on the latest version of software, easily scales as their business grows and provides the flexibility to add functionality (e.g., advanced supply chain management, product lifecycle management [PLM], etc.) as their business complexity expands. Yes, this takes some work and courage to dump what you have, but it definitely provides a much more secure future.”
“Redundancies in software is all too common a problem these days,” says Intelex’s Dea. “While one of the primary benefits of SaaS is the ease with which new services can be turned on, that ease of implementation can also be a double-edged sword, leading some organizations to have significant sprawl and redundancy in tools.”
ERP, MES, QMS?
Whether or not an MES or ERP system can fulfill the needs of quality and/or food safety will depend specifically on its available modules. Obviously, a quality or food safety software solution will be fine-tuned to quality and food safety, and won’t pretend to have the capabilities of ERP or MES. Each system has its place, and the important issue is that they all “talk” to one another.
“The fundamentals for food safety, quality and efficiency should be included in any ERP that is specifically designed for food and beverage manufacturing,” says Infor’s Edgett. “These may be optional modules, but they should be seamlessly integrated.” Examples include a QMS for tracking records, a PLM to manage formulas to ensure label and regulatory compliance and a track-and-trace engine. “All of this is still going to come up short if you don’t have the planning and scheduling tools with process manufacturing variables available to drive efficiency,” says Edgett.
“A recipe-based ERP system that is designed for food manufacturers, along with an MES with real-time information and an EAM system to improve reliability and decrease downtime, can fulfill the necessary requirements for food safety, quality and efficiency,” says Aptean’s Payne. To meet a food manufacturer’s specific needs, the overall system will need to offer bidirectional traceability, recipe management, inventory control and quality management as well as financials, sales and manufacturing modules, adds Payne.
“MES plays a crucial role in handling food safety and quality,” says Rockwell’s Kammer. “Most food safety and quality issues are caused by deviations during the production process. The only way to address these concerns after production is through sorting and containment—that’s often too late. Since MESs drive the workflow of the production process itself, they can include safeguards that keep problems from occurring in the first place.”
A good example is ingredient verification. Material is scanned before the addition to a batch. If it’s incorrect, there is an immediate notification—as oppossed to finding lab results that show the finished product to have the wrong ingredient or too much or too little.
Software tools are designed for specific purposes, e.g., you’d use Word to write an article or Excel to do a spreadsheet and crunch numbers.
“ERP and CRM systems were not designed for full chain digital traceability,” says Trace Register’s Heggelund. “Specialized traceability solutions are needed for those purposes. Each system is designed to meet very specific needs. MESs are typically good at handling internal traceability; ERP systems are traditionally focused on the ‘back office’ task centered around accounting and resource management. Linking external traceability data, provided by traceability systems, with MES and ERP is required to fully manage food safety and quality.”
“Typically, an MES cannot gain true learning from the quality data it records because of a limited ability to perform analytics of trends, correlations and comparisons,” says InfinityQS’s Voight. “In our experience, they tend to be unduly cumbersome with confusing user interfaces that put undue strain on operators.”
While ERP systems are valuable tools for modern manufacturing, they have a couple of shortfalls. They lack the ability to gain insight from quality data by analyzing trends, correlations and comparisons, and they can’t provide real-time feedback and alarming across the management chain to minimize the time required to recognize and resolve issues, adds Voight.
“Data is critical when managing FSQA operations,” says SafetyChain’s Bernkopf. Ensuring data is collected, analyzed and acted upon as quickly as possible is requisite for the highest level of food safety and quality. “Relying on other tools not designed for agile data collection or analysis—which are cumbersome to pull or share data with FSQA team members—does not promote efficiencies that FSQA technologies are designed to achieve,” he adds.
For the food safety/quality team members, MES or ERP systems simply do not fulfill the day-to-day operational needs of FSQA operations, says Bernkopf. “However, with data being critical throughout an organization, FSQA technologies should be able to share data fluidly among other key systems utilized at companies, such as ERP and MES systems.”
Some ERP systems such as SYSPRO have integrated quality systems that provide for the creation and tracking of corrective and preventive actions (CAPA). Benadretti points out the ways that SYSPRO supports HARPC by identifying weak spots in the controls, identifying ineffective controls, identifying new hazards, performing necessary steps to reduce the likelihood of recurrences, evaluating the processed food for safety and preventing adulterated food from entering commerce.
Moral of the story: Pick and choose carefully, matching potential software offerings to your needs now and into the future. If you’ve established HACCP/HARPC programs and are trying to run them on paper, take a look at the software you have now and see if modules can be added, or if you need a more rigorous FSQA system for risk-based products, there are plenty of solutions available in whatever architecture and price range you need.
Making the conversion to electronic
Steven Voight, application engineer for InfinityQS, lists four major steps for converting to InfinityQS’s quality management system, but these general steps will apply for most software applications when you make the change from paper:
- Standardize naming conventions: Work through the various permutations of testing protocols, naming conventions and specifications that almost certainly exist in the company. Simplify to the max. Some variations are valid and necessary, but many will be arbitrary.
- Select the deployment method: Deciding on an implementation architecture for the software solution is critical. If it’s necessary to get up and running quickly with minimal IT structure, then a cloud solution may be in order. But, if there’s already a robust and well-maintained infrastructure in place, an on-premise solution may be preferable.
- Digitize records: Convert the existing paper forms to electronic. While it can be daunting, working closely with the engineers from the software provider can ease the task.
- Instill change management: The last hurdle will be to get everyone using the software properly and taking full advantage of its features and functionality. Creating a culture of quality and instilling proper change management initiatives will smooth the way to full usage and benefit.
For more information:
Maryanne Steidinger, Schneider Electric, 877-342-5173,
Gordon.benzie@schneider-electric.com, software.schneider-electric.com
Jon Simpson, Sage Clarity, 800-809-3042,
jon.simpson@sageclarity.com, www.sageclarity.com
Dan Bernkopf, SafetyChain, 888-235-7540,
info@safetychain.com, www.safetychain.com
Jack Payne, Aptean, 770-351-9600,
jack.payne@ aptean.com, www.aptean.com
Jason Dea, Intelex, 416-599-6009,
Jason.dea@intelex.com, www.intelex.com/industries/food-beverage
Dag Heggelund, Trace Register, 206-621-1601,
dheggelund@traceregister.com, www.traceregister.com
Steven Voight, InfinityQS, 800-772-7978,
sales@infinityqs.com, www.infinityqs.com
Mike Edgett, Infor, 800-260-2640,
mike.edgett@infor.com, www.infor.com
Joey Benadretti, SYSPRO USA, 714-437-1000,
info@us.syspro.com, www.syspro.com
Scott Deakins, Deacom, Inc., 800-433-2266,
kpeters@deacom.com, www.deacom.com
Khris Kammer, Rockwell Automation, 440-646-3434,
kkammer@ra.rockwell.com, www.rockwellautomation.com
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!








