Imported Ingredient safety
How safe are imported ingredients?
FDA, USDA, audits, COAs, independent testing and labeling all help to keep our foods safe

U.S. food manufacturers and retailers look outside of the country to increase the food supply. As of 2019, the U.S. imported about 15% of its overall food supply. Importing human food ingredients might sound like a no-brainer: that’s what the FDA is for, after all. Yet in reality, it is not that simple. Photo courtesy of Getty Images / Aamulya
Americans eat a lot of imported foods—and demand is growing with the likes of plant proteins, seafood, new varieties of vegetables and fruits, and even beef, along with simple ingredients such as spices and coconut milk.
To help meet consumer demand, U.S. food manufacturers and retailers look outside of the country. As of 2019, the U.S. imported about 15% of its overall food supply. We don’t really consider the likes of our usual foods as imported—like bananas or even coffee. Importing human food ingredients might sound like a no-brainer: that’s what the FDA is for, after all. Yet in reality, it is not that simple.
The FDA must take steps to protect the public from a threat, or actual terrorist attack, on the U.S. food supply and other food-related emergencies, per the Public Health Security and Bioterrorism Preparedness and Response Act of 2002. Let’s look at another Act under the FDA that explains a little more.
Under the U.S. Federal Food, Drug and Cosmetic Act, the FDA states:
Importers of food products intended for introduction into U.S. interstate commerce are responsible for ensuring that the products are safe, sanitary, and labeled according to U.S. requirements. (All imported food is considered to be interstate commerce.)
FDA is not authorized under the law to approve, certify, license, or otherwise sanction individual food importers, products, labels, or shipments. Importers can import foods into the United States without prior sanction by FDA, as long as the facilities that produce, store, or otherwise handle the products are registered with FDA, and prior notice of incoming shipments is provided to FDA.
Imported food products are subject to FDA inspection when offered for import at U.S. ports of entry. FDA may detain shipments of products offered for import if the shipments are found not to be in compliance with U.S. requirements. Both imported and domestically-produced foods must meet the same legal requirements in the United States.
The FDA also states that food importers must have a U.S. food agent acting as a liaison for FDA communications regarding incoming food shipments. And, that they will communicate with that agent to schedule inspections and verify that all food safety requirements are being met.
But is that happening? There are various programs for importers and the facilities that accept them, third-party certification programs, audits and more.
What it comes down to, is that U.S. manufacturers must be not only aware of the FDA’s responsibility, but also be proactive with their own food safety management system, certificates of analysis, audits, SQF (safe quality food programs) and so on. Because of course, these facilities will be on the hook for ramifications of the foodstuffs going onto Americans’ tables.
 Food labels identify ingredients, including imported, in the products we eat. Photo courtesy of Getty Images / Hailshadow
Food labels identify ingredients, including imported, in the products we eat. Photo courtesy of Getty Images / Hailshadow
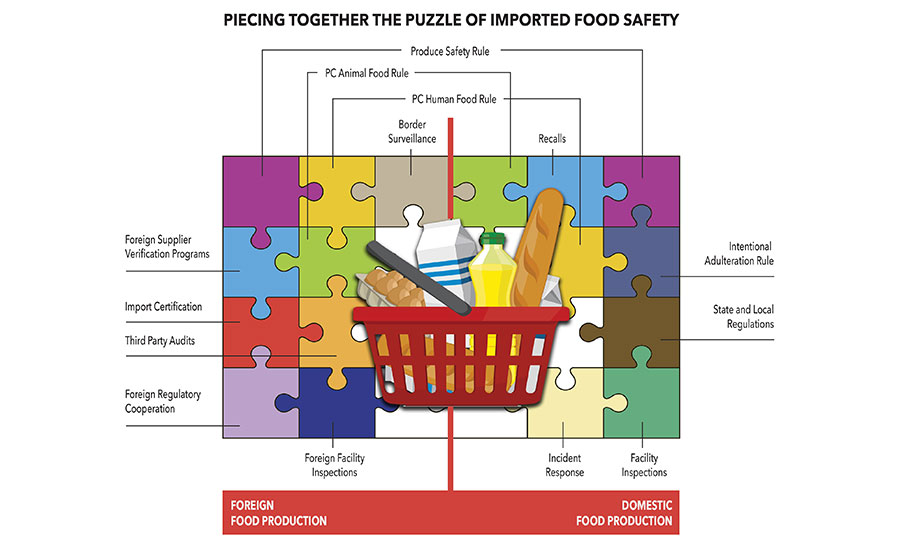
Foreign Supplier Verification Programs
One of the FDA programs leading the charge on imported food safety is The Foreign Supplier Verification Program. Its goal is to minimize the risk of Americans contracting a serious foodborne illness or encountering other safety problems from contaminated food.
The FSVP final rule makes importers accountable for verifying that foreign suppliers are producing food in a manner that meets U.S. safety standards. The rule requires that importers of food to the U.S. are using the most appropriate verification activities to significantly minimize or prevent risks and prevent adulterated foods from reaching America’s dinner tables, as well as foods that are misbranded with respect to allergen labeling. Importers are also required to verify that their suppliers are producing food in a manner that provides the same level of public health protection as required under the other new FSMA regulations on preventive controls and produce safety. The verification activities could include annual onsite audits of a supplier’s facility, sampling and testing of food, or a review of the supplier’s relevant food safety records.
The Foreign Supplier Verifications Programs rule, in effect as of January 2016, requires importers to perform risk-based foreign supplier verification activities to verify that:
- The food is produced in a manner that provides the same level of public health protection as section 418 (concerning hazard analysis and risk-based preventive controls) or 419 (concerning standards for the safe production and harvesting of certain fruits and vegetables that are raw agricultural commodities (RACs) of the FD&C Act (21 U.S.C. 350g and 350h), if applicable;
- The food is not adulterated under section 402 of the FD&C Act (21 U.S.C. 342); and
- The human food is not misbranded under section 403(w) of the FD&C Act (21 U.S.C. 343(w)) (concerning food allergen labeling).
This past May, the FDA launched the FSVP Importer Portal for records submission so importers can upload records electronically to the agency after receiving a written request from FDA. Currently, the FSVP List includes more than 25,000 companies.
 Click image to enlarge
Click image to enlarge
Meat & Poultry
Regarding meat and poultry, under the USDA, The Food Safety and Inspection Service (FSIS) is responsible for inspecting all raw meat and poultry sold in interstate and foreign commerce, including imported products. The Agency monitors meat and poultry products after they leave federally inspected plants. Products must be safe, and properly labeled and packaged.
The FSIS Import & Export Library identifies countries that have been determined, through the USDA, Food Safety and Inspection Service (FSIS) Equivalence Process, to have equivalent meat, poultry or egg product food safety inspection systems are eligible for import. The Library also identifies the meat, poultry or egg products each equivalent country is eligible to export to the United States. The eligible products for each country are identified using the Process Category, Product Category and Product Group listed in the FSIS Product Categorization document and utilized in the Public Health Information System (PHIS). They are outlined as:
- Process Category: High-level category based on whether and how products are processed after slaughter
- Product Category: More specific category that links the product to the appropriate species and provides more information on the production process
- Product Group: Most detailed description of eligible products used by FSIS to program appropriate types of inspections in the FSIS PHIS
Additionally, the USDA Animal and Plant Health Inspection Service (APHIS) restricts certain animal products from entering the U.S. because of animal disease conditions in the country of origin.
However, if the food contains a small amount of poultry, eggs or meat, The Federal Meat Inspection Act, the Poultry Products Inspection Act, and the Egg Products Inspection Act and the FSIS implementing regulations allow FSIS to exempt certain foods, provided that the meat, poultry or egg product component of these products is from an approved or eligible source.
For meat and poultry, the FSIS policy is:
In the case of meat, the “relatively small proportions” of meat ingredients are:
- 3% or less raw meat;
- Less than 2% cooked meat or other portions of the carcass; or
- 30% or less fat, tallow or meat extract, alone or in combination.
In the case of poultry, the “relatively small proportions” are:
- Less than 2% cooked poultry meat;
- Less than 10% cooked poultry skins, giblets or fat, separately; or
- Less than 10% cooked poultry skins, giblets, fat and poultry meat (limited to less than 2%) in any combination
 Food manufacturers should have in place a food safety management system, along with audits and/or certificates of analysis, in order to be compliant.Photo courtesy of Getty Images / vusta
Food manufacturers should have in place a food safety management system, along with audits and/or certificates of analysis, in order to be compliant.Photo courtesy of Getty Images / vusta
Guidance for imported food safety
EAS Consulting Group is a leading provider of regulatory compliance solutions to industries regulated by FDA, USDA and supporting agencies. The EAS team of consultants provides expert guidance for meeting the necessary regulatory requirements for various industries including food, dietary supplements and cannabis.
Susan Moyers, independent consultant, food and dietary supplements at EAS Consulting Group, shared some insights on how the company works with food manufacturers in the areas of food safety, food defense, food labeling, operational and formulation, ingredient and packaging programs to support FDA and USDA requirements and determine regulatory risk. EAS Consulting assists importers in meeting the standards of the Foreign Supplier Verification Program (FSVP) and other regulations. And also coaches companies on meeting private industry standards.
In terms of certification bodies, Moyers says that there are non-governmental organizations with provisions that subscribing companies have to be compliant with, but they do not have a governmental enforcement power.
The most well-known credentialing organization is the Global Food Safety Initiative (GFSI), headquartered in Paris. “The goal of this group is to create standards for retailers, so it’s a retailer-driven non-governmental standard that most of the private audits that occur are conforming to,” says Moyers.
The GFSI started around the year 2000, and The British Retail Consortium was a part of that; and the most popular one in the U.S. is called SQF, Safe Quality Food. “The board of the GFSI consists of large retailers and large producers – Coca-Cola, Amazon, Walmart have had members on the board,” explains Moyers. “The GFSI creates benchmark standards, and then organizations such as the Safe Quality Food Institute (SQF) and the British Retail Consortium (BRC) use those benchmarks to develop their respective audit programs. They then authorize various certification bodies to conduct audits against the program. Auditors in the field are working under the rules one of the certification bodies using programs developed by SQF, BRC and similar standards organizations, such as ISO. The ISO equivalent food safety program is known as the Food Safety System Certification, or FSSC22000.
The GFSI standards are not governmental. However, most large retailers accept these private standards as part of their overall food safety plans. Moyers says that if a food manufacturer is not ISQF or BRC-certified, some retailers just won’t do business with you.
Actually, the BRCGS (global standards for BRC) has been adopted by over 20,000 sites in 130 countries, and the standard is accepted by 70% of the top 10 global retailers, 60% of the top 10 quick-service restaurants and 50% of the top 25 manufacturers.
The difference between ISO and BRC standards is that the BRC standard has a focus on quality, food safety and legality. SQF has separate audit programs for food safety and food quality. The FSSC 22000 targets food safety and legal compliance.
So what about our own FDA or USDA? With regard to the USDA for meat and poultry, everything is required to be inspected. Moyers says that the FDA does get involved, but it depends on the product. “Under FSMA (Food Safety Modernization Act), there is a regulation called ‘intentional adulteration’, so under that umbrella, companies who comply with FDA standards have to look at some types of food fraud, as FDA looks at food fraud that impacts safety, i.e. intentional adulteration such as poison, terrorist event, etc.
“So they’re not looking at FDA for food fraud for quality – they’re looking at fraud in terms of safety through food quality,” Moyers says. However, some foods have standards of identity to be used in food commerce, such as milk chocolate, cheddar cheese, all dairy products. The FDA has enforcement power that if a product calls itself cheddar cheese and it isn’t, and doesn’t meet the food’s standard of identity, then the FDA will be concerned.
Moyers goes on to say that, over the years, one of the FDA’s biggest concern has been seafood, due to mislabeling. Unethical suppliers will take a cheaper cut and call it a more expensive cut of fish, for example. Globally, there is now DNA typing for all of the 35,000 plus species. For out of U.S. fisheries, they must have an MOU, a memorandum of understanding, in order to bring your seafood into this country.
The most controversial issue for standards of identity is olive oil with so many substitutions with oils out there. “An olive oil group from California is trying to get the FDA to get a standard of identity for the safety of their products. There have been scandals with oils coming out of Italy. There have been petitions sent to the FDA for years but nothing has been done yet,” explains Moyers.
FDA has jurisdiction and enforces food safety as part of the FSMA, as far as quality goes, it’s only certain products that have standards of identity for quality, she adds.
So what can we do? Many larger manufacturers have their own safety protocols in place. And use the GFSI as a baseline. They may also have an audit team to perform a separate customer audit.
A Certificates of Analysis is a document that comes from the manufacturer designed to attest to the quality and/or safety of a product or ingredient. But there is no uniform document for COAs—some list specific things like taste, texture and color while many talk about microbiological contaminations. With regard to COAs, I always think about what Ronald Reagan once said: ‘trust but verify.’”
“With regard to COAs, I always think about what Ronald Reagan once said: ‘trust but verify.’”
— Susan Moyers, EAS consultant
One provision in the FSVP states that if a foreign supplier doesn't control a certain hazard, they have to have a supplier-approval program (for ecoli, etc.) and have an annual audit. “Only foods with hazards you can’t control, i.e., produce and ingredients you’re not cooking so you can’t control the microorganism. You’re not controlling what’s going on, you’re relying on your supplier for that food safety,” Moyers explains.
This puts the impetus on importers to verify their suppliers overseas meet U.S. standards. Any ingredient supplies preventive control rules in FSMA. All suppliers to the U.S. must meet the standards.
Manufacturers should also consider independent testing on ingredients. Some have internal labs and some will send out testing to independent certified laboratories. “I believe it’s a good idea to do periodic checks,” Moyers says. Some countries have been less compliant and the companies have to be vigilant to verify food safety and fraud.
The issue with this verification program comes from brokers, etc. who don’t know anything about food safety. Wholesalers, brokers and dealers this affects most. There is a lot of misunderstanding. Each segment of food products have different standards. It requires understanding of FDA regulations to service a wholesaler or dealer adequately.
Moyers offers some tips for manufacturers who are unsure of regulations or new companies: Consider the GFSI certification. Maybe do an independent audit of supplier before engaging. Some companies do periodic testing against specifications and safety parameters using accredited labs. Testing and education are important, Moyers adds. “When we do an audit, you say what is. When consulting, you can give advice… Is there a weakness in the program that we will advise against.”
Smaller companies may not be aware of these qualifications, so education, even EAS webinars, FDA documentation, etc. A company should be familiar with Foreign Supplier Verification Programs, standards of identity.
For more information visit easconsultinggroup.com, fda.gov and usda.gov.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!





