Risk-based Sanitation
Develop a Risk-based Approach to Sanitation
Having a plan and taking action on a continual basis helps keep uninvited squatter bacteria out of your facility.

Rachel McGinness, lead chemical engineer at Ecolab, completes a titration via test kit in a small dairy plant to check CIP effectiveness. Image courtesy of Ecolab
This is the not the type of letter a food processor wants to receive from FDA:
Your food safety plan for RTE Thai Noodle Salad and Macaroni and Cheese did not identify a preventive control for the food safety hazard of contamination of RTE products with an environmental pathogen, e.g., L. monocytogenes. These RTE products are exposed to the production environment prior to packaging and the packaged food does not receive a treatment or otherwise include a control measure. Furthermore, WGS (whole genome sequencing) analysis revealed two (2) different strains or clusters of L. monocytogenes that have become persistent in your facility.
Persistent or “resident” bacteria of any kind in a food plant—and especially listeria—typically means a facility and its equipment haven’t been cleaned properly, and while there might be an FDA-based HACCP (hazard analysis and critical control points) and/or HARPC (hazard analysis and risk-based preventive controls) plan in place for regular and thorough cleaning and sanitation, it’s not being diligently followed. In some extreme cases, facilities with resident listeria that couldn’t effectively be removed by repeated cleaning and sanitation have been shut down and/or razed.
How can processors establish an effective risk-based cleaning and sanitation plan that not only ensures proper cleaning of all spaces in a food plant, but also satisfies regulatory requirements as well? What should a risk-based sanitation program look like, and how can it pass muster with FDA, USDA and other regulatory authorities?
“Ecolab recommends a multi-step approach to risk mitigation,” says Rick Stokes, manager, area tech support. The chemical company has worked with thousands of manufacturers to uncover potential risk areas such as inadequate sanitation methods, food safety issues, poor water quality, etc.

“The following steps—risk assessment, risk prioritization and risk mitigation––are where the rubber meets the road,” says Stokes. “We identify the most urgent issues based on our industry expertise and formulate a game plan for mitigating or eliminating risk, which sometimes involves hands-on training or sanitary design consultation. The final step, which is ongoing monitoring and adaptation, is crucial. This is where we can use sophisticated digital tools to drive ongoing, targeted service based on real-time analysis and custom sanitation plans.” Overall, Stokes recommends a proactive stance on risk management.
Architects and engineers are a good source of help in finding and preventing food safety issues arising from microbiological organisms. “Good sanitation and cleaning practices start with integration of thoughtful design practices of the facility,” says Enrico Chua, associate senior architect for Gresham Smith. Sanitation and cleaning operations are an integral part of Gresham Smith’s architectural and engineering design practice, and the firm has leveraged its years of experience by employing a best-practice approach through lessons learned not only with food and beverage companies but from other markets that address similar challenges. These include maintaining optimal cleanliness and managing critical and tight environmental controls—whether designing new facilities or retooling, renovating and adding on to existing facilities.

Gray Solutions, a Control System Integrators Association member, works closely with food manufacturers to understand what their process hazards are, and how to mitigate those hazards through effective cleaning, says Adam Richards, food safety engineer. “Most of the processes we work in will use clean in place (CIP) to assist with their cleaning efforts. Clean-in-place systems help from a labor standpoint, and the turnaround time is typically quicker. We can assist the customer with chemical dosing station recommendations and what cleaning chemicals are best for their process.”
Many Gray Solutions’ employees have completed hygienic design training and understand a processor’s needs regarding what cleaning chemicals and building materials would be best for specific areas, says Justin Basham, commissioning engineer. Gray can also help with choosing equipment best suited for the area, placement of the equipment and elimination of pathogen harborage points—including the correct stainless-steel grade to prevent corrosion.
Tools and Training are Lacking
Problem is, however, many U.S. plants are simply aging, says Ecolab’s Stokes. “They aren’t set up with the equipment and facility designs they would need to invest in modern monitoring tools, and without those tools they lack insights that support better efficiency, food safety, team capabilities, etc.”
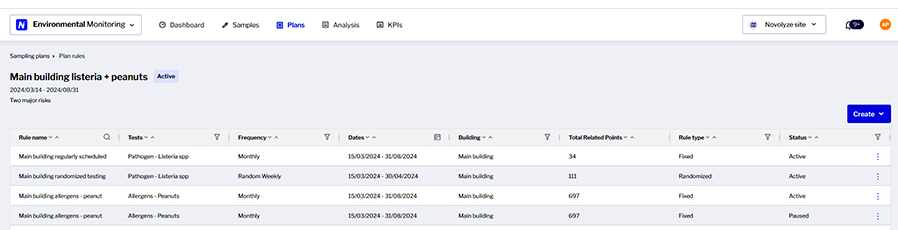
One set of tools, the Novolyze platform, aims to increase food safety and quality performance by offering software modules for environmental monitoring, sanitation, smart process control, smart validation and quality forms, says Moussa Ndiaye, product manager. The Novolyze Sanitation Complete module provides digital tools to build a risk-based master sanitation schedule that will take into consideration any risk that might occur in a given factory. The system also supports task force shortage and turnover by facilitating onboarding and training.
“Some facilities aren’t taking advantage of modern training programs, and I think a big part of that is simply not knowing how good training has become,” adds Stokes. “The level of training your team can receive is phenomenal if you know where to look for it, and it can make a measurable difference in your processes.”
Training employees so they can understand what hazards are associated within each department is crucial to a clean facility, says Gray’s Richards. An example of this would be someone working in a raw meat area having to walk through a finished-goods area with open product contact zones. The likelihood of them contaminating the product may be low, but the severity could be detrimental to the product.
Training should be ongoing to be effective. “Along with initial training of employees, annual or bi-annual refresher courses associated with food safety concerns tend to keep it in the forefront of everyone’s minds,” adds Gray’s Basham. The use of practical “quizzes” during the initial and refresher courses gives the employees a way to see how the decisions they make will affect the quality of product they send to customers.
Even if given adequate training, workers will find that some facilities make cleaning inherently difficult. “Often, a facility’s building design is disconnected from its sanitation and cleaning plans due to being developed independently,” says Gresham Smith’s Chua. “A more effective approach integrates both aspects. This becomes very apparent when acknowledging that environmental contamination is a major source of the contamination of processed foods.”

HACCP, HARPC or Both
HACCP has been around for some time, but HARPC (hazard analysis and risk-based preventive controls) came out of FSMA. What is the better approach with cleaning and sanitation?
“The answer depends on the regulatory and/or customer requirements in place,” says Stokes. “For most processors, this choice is determined by the product that is being made. Regulations may require HACCP or HARPC approach.”
If a process has a relatively narrow product exposure window, and if the sanitation is controlled through very repeatable means like CIP or closely inspected after every cleaning, then a manufacturer may not see as many benefits in the HARPC philosophy, adds Stokes. “But I do see advantages of the HARPC approach to systems that have a wide exposure window or limited treatment to address potential hazards. In this case, HARPC offers an opportunity to really explore the risks along the entire chain from supply to consumer.”
“If HACCP is used as intended, I think you can create an effective sanitation and environmental monitoring program without adding HARPC,” says Richards. The first step to HACCP is creating the cross functional team—people from all different positions in the facility so you have extra eyes on the process. Once you have a team in place, then you can start to discuss the hazards and how you plan to control them—whether that’s through control points, cleaning or personnel training. To create an effective cleaning program, you’ll need to collect data to prove that your cleaning procedures are effective. This means swabbing the equipment and facility before and after cleaning to understand if you’re getting the equipment clean and free from microbial growth. You can then use that data to track and trend when you do have a presumptive positive in a specific area.
“Integrating the principles of both HACCP and HARPC would be a better approach as it recognizes the interconnectedness of product and environmental risks and addresses them together,” says Chua. “This also provides clear direction to all stakeholders at all stages of the project’s life cycle.”
A Risk-based Approach to Sanitation/Cleaning
How can processors develop an effective approach to sanitation/cleaning and hygiene that prioritizes the right areas of the plant and/or equipment to be cleaned and meet regulatory rules?
“Identify the risks and their severity within each area of the facility,” says Gray’s Basham. Develop a schedule of cleaning based on the severity of the risks within that area, cleaning the areas that pose a greater threat more often or with more stringent practices than those that do not. Understanding the types of pathogens that could be present in different areas of the facility and the best approach to their mitigation is paramount. Using a wet cleaning method in a dry area will most times do more harm than good.
It all comes down to effective monitoring and better use of sanitation zoning, says Ecolab’s Stokes. You can set up your cleaning methods and tools as rigorously as you want, but without attentive monitoring and meaningful adaptation to data they’re not going to make the impact they’re capable of making. It’s important to begin with a thorough understanding of what you’re monitoring for and where you can find that information, and the results of that data collection will feed back into a formalized review process that helps you continuously reestablish your risk profile.

“My advice to customers is this: Treat regulatory rules as a minimum bar,” adds Stokes. “Develop an effective sanitation program that addresses the full spectrum of your sanitation-related risk. Your company is the expert when it comes to what you make and how you make it, so direct that deep knowledge toward creating a superior sanitation program (complete with employee training and effective tools) that thoroughly meets the site-specific requirements for keeping your product safe. Then, review regulatory requirements and other sanitation-related standards to ensure that your program hasn’t left any gaps covered by specific regulation.”
And don’t forget metrics. “Identifying measurable metrics against risk exposure and probabilities can help food processors develop an effective risk-based approach to sanitation and cleaning,” says Chua. “Metrics allow for focused targets versus vague ideals, therefore providing a baseline and point of verification for effectivity.” An environmental monitoring program can establish procedures and methods in cleaning, sampling, analyzing and trending.
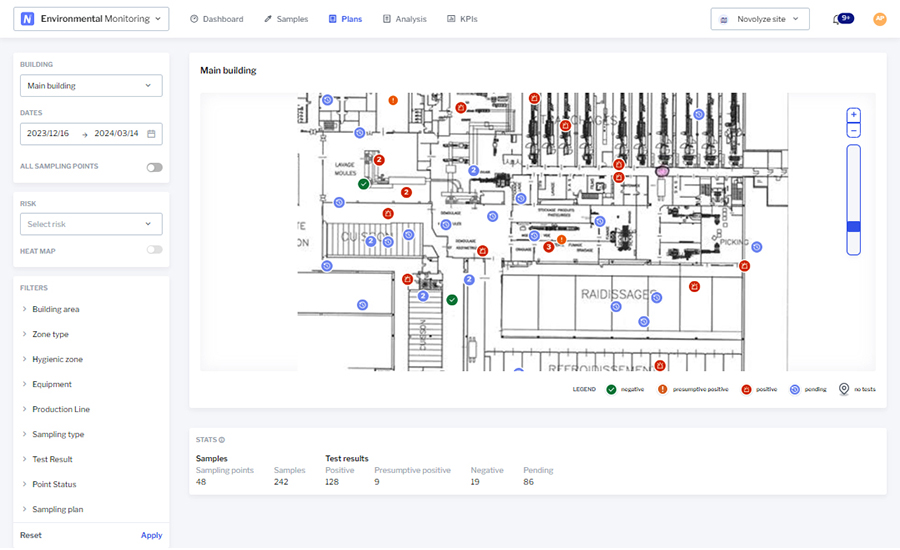
“Understanding risk requires strong analysis and data supporting risk identification,” says Ndiaye. “Data visualization tools, such as those offered by Novolyze Environmental Monitoring and Sanitation Complete, are necessary to spot worst case areas and equipment.”
Resident Bacteria: Eradicate or Demolish the Building?
Sometimes resident bacteria so permeate a facility that it’s impossible to get rid of the bugs without razing the structure. I can remember one or two such cases. But it doesn’t have to be this way.
“Razing the entire facility or a ‘scorched earth’ approach, although an option, should be a last resort,” says Gresham Smith’s Chua. “There is still hope in eradicating the bacteria if there is a clear methodology to isolate the incidence and understand how it occurred. In most cases, this clear path is through an environmental monitoring program with sampling, testing and analysis.”
Think about how bacteria can become resident bacteria, says Novolyze’s Ndiaye:
- Adaptation to chemicals—bacteria can adapt to chemicals and stress
- Shielded location—bacteria accumulate in hard-to-reach areas
- Ineffective sampling plan—sampling in the wrong locations; sampling plans need to be adaptive and evolve over time
Changing chemicals regularly and efficient mapping of sampling plans can support identifying bacteria in shielded areas, adds Ndiaye.
“Applying strong sanitation principles designed to limit or remove bacteria will often remove resident organisms,” says Ecolab’s Stokes. “I’ll add that a robust understanding of microorganisms and the tools used to detect them is essential.”

“It’s tempting in these situations to look for a ‘smoking gun’––a single source or trigger for the problem, says Stokes. But looking at it this way may miss the root of the problem. More often than not, it’s not a single source but a network of poor conditions that make resident organisms possible. I have seen many companies target the symptoms instead of attacking the root of the problem, and this approach rarely yields positive results.”
Typically, you would rely on your swab data to track where the listeria hits are showing up and do a ‘swabathon’ in adjacent areas to see where it’s going,” says Gray’s Richards. [1] “Once you have a map of hotspots, you can start with a deep cleansing of those areas, while minimizing foot traffic to avoid tracking it into other areas. After you have effectively cleaned all contaminated areas, continue with another swabathon to ensure that the areas can resume production. Once production has returned to normal, the food safety team should do a corrective action report to understand how the listeria was able to form and why it wasn’t detected before it got out of hand. The cause could be due to the cleaning schedule and not being cleaned as frequently as needed, but I have found that training is usually the number one cause of listeria or salmonella outbreaks.”
Sources:
[1] “What is a Swabathon?: Understanding the FDA Investigations,” Mérieux NutriSciences, (July 13, 2017) Website accessed 3-29-2024
“Foodborne Biological Hazards: Sampling for foodborne biological hazards, and filth– domestic and import,” FDA Compliance Program Guidance Manual, FDA (6-03-2022)
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!







