Engineering R&D
CERTUS helps processors simplify Listeria testing
This user-friendly system provides real-time monitoring through simultaneous enrichment and detection.

The CERTUS System provides straightforward and simple pathogen testing in three easy steps in a fully bio-contained system. Source: CERTUS.
Running a microbiological test to detect Listeria present in a plant has always been problematic. If it couldn’t be run in house, it was sent to a lab with results provided days later. If testing equipment was available, experienced operators had to run it, and it still took 24 hours or more to get a result.
A startup company has purchased licensing rights to a new technology and built a system that allows anyone to take samples and run a Listeria test. The company, CERTUS, has constructed a hardware-software system that simplifies testing for Listeria, providing quantitative results in real time. Besides environmental testing, the analyzer will also be able to test for Listeria, Salmonella and E. coli in food samples. Environmental monitoring will be the initial rollout for Listeria and Salmonella, followed by E. coli. Later, plans for food testing will be introduced to the market.
The CERTUS pathogen detection system was designed and developed through strategic collaborations, says President John Coomes. The core SERS (surface enhanced Raman spectroscopy) technology and instrument design were licensed exclusively from Becton, Dickinson and Company (BD) for applications in food safety. Assay reagent development has been conducted in collaboration between CERTUS and UK-based Solus Scientific. Solus has expertise in the development of immunoassay-based tests, specifically for food pathogen testing, and has a line of AFNOR- and AOAC-validated ELISA tests.
Coomes has big plans for migrating this technology beyond environmental Listeria, but most of all, he wants it to be affordable for medium-sized processors, offsetting all the costs involved in farming out tests to a local lab. Coomes describes this technology and explains how it’s different from other methodologies.
FE: How long has this system been in development?
John Coomes: The technology was licensed from BD in January of 2016. Since then, full-scale development has been underway on the instrument, test kits and software.
FE: How is this system different from other methodologies on the market?
Coomes: The CERTUS System is unique in its simplicity and ease of use. There’s no need for startup kitsor ancillary equipment, such as [a] centrifuge, pipettes, etc. No sample preparation is needed, and anybody in the plant from the sanitation crew to CEO can run the test with virtually no training.
The CERTUS System includes everything needed for testing—the actual machine, called the detection unit, consumable test kits and the control pad, which not only guides the testing process, but also logs and records all data for FSMA compliance.
Another difference is the ability to send presumptive positive samples to a third-party lab for confirmation, if needed. Test methodologies that employ genetically engineered simulations of bacteria make such confirmations impossible.
The CERTUS Grow, Read, Detect process is unique in its ability to detect pathogens during incubation. Many other systems require full incubation of up to 24 hours before analyzing for the presence of pathogens.
FE: What is the typical workflow?
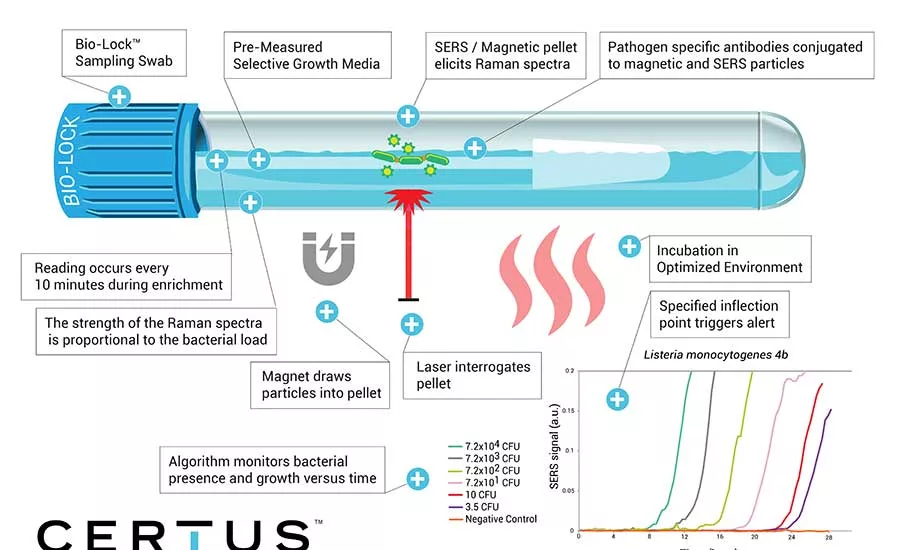
Coomes: It’s pretty basic compared to some systems. First, use a Bio-Lock sampling swab on the suspected surface. Next, add media that comes premeasured straight out of the detection kit. Then, close the Bio-Lock cap, insert into the detection unit and walk away. The machine simultaneously grows, reads and detects any pathogen, alerting predetermined contact people via email and text.
FE: You say the system provides results very quickly. How quickly? What allows it to provide fast turnaround times?
Coomes: For high-load samples, our current data is showing results in eight to 12 hours. We continue to optimize and strive for shorter times. In BD’s research, much shorter times were achieved with over 20 different food matrices. The fast results are related to the fact that the detection of the pathogens happens from the very instant that you begin to enrich the sample. Simultaneous monitoring occurs through Grow, Read and Detect process steps.
FE: In a nutshell, explain how the system works.
Coomes: The CERTUS System is a simple, safe smart detection system. It starts with a three-step workflow that eliminates extra equipment. Using a SERS detection method, the system continuously monitors and identifies growing pathogenic bacteria without negatively affecting growth.
Embedded in the bottom of each detection tube are pathogen-specific dry-down particles, which, when combined with the premeasured growth medium, conjugate pathogen-specific antibodies to magnetic and SERS particles. Once a sample is collected, sealed in the Bio-Lock detection tube and inserted into the detection unit, enrichment and detection begin immediately. The detection unit features a thermal chamber optimized for growth of the selected pathogen. Occurring at regular intervals, a magnet is deployed, pulling the particles into pellets, which are then interrogated by a laser. The Raman spectra elicited by the pellets is read, logged and applied to a logarithm that monitors bacterial presence and growth versus time. A specified inflection point in the algorithm triggers an alert, which can occur at any time throughout enrichment.
FE: Can the system test for other foodborne bacteria?
Coomes: Not right now, but building on preexisting R&D from BD, CERTUS plans to offer multiple tests, such as Salmonella, E. coli and Campylobacter for both environmental samples and end-product food samples.
FE: Some methodologies involve a lot of handling and can expose workers or the plant to grown bacteria. How does CERTUS protect workers from accidental contamination?
Coomes: The CERTUS System is fully bio-contained. Our Bio-Lock sampling swabs use technology to completely seal collected samples within the detection tubes. Once the Bio-Lock caps are screwed closed, they cannot reopen. When testing is complete, the entire detection tube is disposed of.
FE: How does this system compare in cost to sending samples to a lab? How long would it take to pay for itself? What are the costs of any expendables?
Coomes: The total cost of the instrument, iPad and test kits will be competitively priced, and the total value of the efficiencies gained will be attractive to medium-sized food producers.
FE: Are you currently testing this device with any food and beverage companies? If so, what have been their impressions?
Coomes: Combined with extensive market research at food and beverage processors, test labs and government bodies, CERTUS has done additional voice-of-customer research. In all cases, the simple, safe and smart approach offered has been very well received. Beta testing at sites with the new system are scheduled for late fall of this year.
FE: What approvals do you already have or are still waiting to get?
Coomes: AOAC submission preparation is underway now, and we have been engaged with AOAC on this front for several months now. CERTUS is an AOAC member.
FE: When will CERTUS Systems be available to the industry?
Coomes: CERTUS Systems will be available in early 2018.
For more information, visit www.certusfoodsafety.com.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!






