Food Safety
Manage recalls with the tools you already have

A single-system ERP architecture can manage incoming ingredients and place a QC hold on them until they are deemed fit for production. Likewise, when the product is finished, a QC hold can be placed on shipping it until QC tests are completed. Source: Deacom.

The convergence of operation and information technology helps provide contextualized data that relates to process conditions. Source: Rockwell Automation.

The InfinityQS International enterprise quality management solution provides a lot traceability and genealogy function to help users track raw material codes throughout their manufacturing operations. Source: InfinityQS.
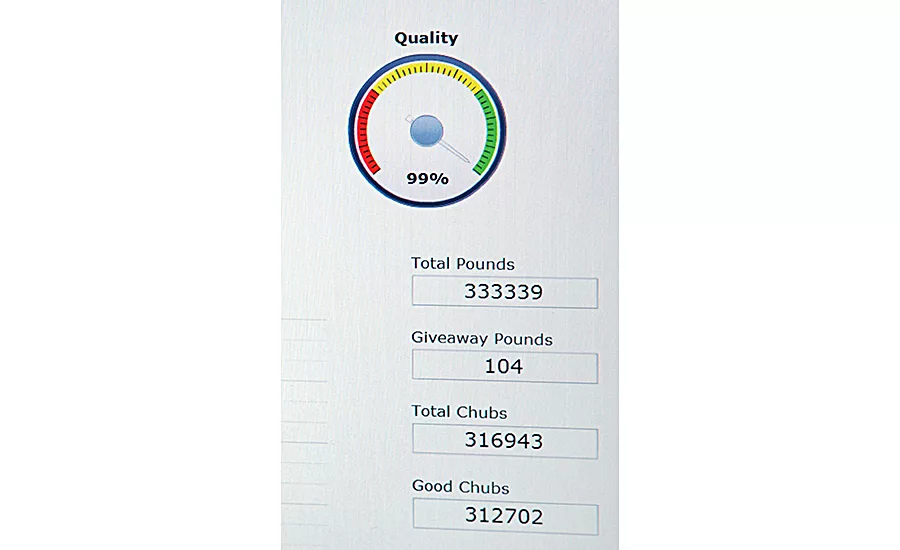
Hillshire Brands monitors about 1,500 data points across cooking, sticking, battering, freezing and packaging. Source: Rockwell Automation.

The Reportable Food Registry (RFR) is now part of the FDANIH Safety Reporting Portal. Reports can be prepopulated with contact information and saved as drafts or partially completed reports. Users also can now view any submissions they previously made to the portal. Source: FDA.
• 1990: Perrier recalls 160 million bottles of water due to benzene contamination. The brand never recovers its prior status.
• 1993: Jack-in-the-Box products are contaminated with E. coli, resulting in numerous injury claims, four deaths and more than $100 million in losses.
• 2008: Peanut Corporation of America ships Salmonella-laced peanut paste, affecting more than 200 companies and 2,000 different products, causing a $1 billion loss to the industry.
• 2013: A false report of botulism contamination results in a $100 million loss for Fonterra in New Zealand.
Granted, these are notorious and expensive recalls, but any recall can be expensive or potentially put you out of business. Your best option: Prevent a recall from happening by having a food safety management system in place. (See “The right tools enable long-term success,” FE, June 2014.)
But, if you must recall product, you need to have the tools that can stop it in the supply chain as quickly and as near your plant as possible. These tools also can narrow the scope of the recalled defective product (that is, reduce the size of your recall) and/or prevent it from getting out the door. Fortunately, you may already have some of these tools. For example, your shop floor manufacturing execution system (MES) and/or enterprise resource planning (ERP) system can receive data from your lab and/or quality systems.
ERP: The big picture
If your ERP is designed for the food industry, chances are, managing recalls will be easier for you. “The food industry is unique in many ways,” observes Joe Scioscia, VAI vice president of sales. “Inventory management, lot and date tracking, quality control and recall reporting are critical.” The VAI S2K Enterprise for Food ERP system is specifically designed to meet these requirements and help processors cope with the challenges of FSMA by automating and integrating the business process across the organization. “A food ERP system offers full traceability while providing comprehensive reports for auditors,” adds Scioscia.
ERP systems should provide comprehensive lot and serial number tracking from raw goods as they are received, through production and into finished goods.
Recalls controlled by an ERP process are normally executed properly, says Jay Deakins, president of Deacom. “The problem is, people bolt together all these different systems, and they get holes in them.”
Consider this hypothetical: QC has checked an incoming ingredient and is waiting for test results before releasing it to production. But, production needs to make a product and has run out of the ingredient. There are no controls in place and no communications between software systems, so the ingredient gets used anyway. When QC has the final test results, the ingredient is found to be defective, i.e., containing an unwanted allergen. In a rush to fill the order, the product has already gone out the door. “It’s a combination of human error, coupled with systemic error, and that’s how you end up with a recall,” explains Deakins.
According to Dan Griffin, director of product management at Exact Macola, ERP systems should provide comprehensive lot and serial number tracking from raw goods as they are received, through production and into finished goods. The Exact Macola Version 10 system has the ability to link manufacturer serial/lot numbers to internally assigned serial/lot numbers so everything is visible through the manufacturing process. “Business process monitoring can have rules that automatically trigger activities if a defect is found during an inspection and send out alerts through the process management system to notify someone that either other lots or serials need to be validated or possibly pulled out of production, preventing tainted food from making it out of the production facility,” adds Griffin.
Prevention must be built into an ERP system, says Jack Payne, Aptean vice president product management and solutions consulting. He believes an ERP system used in the food and beverage industry should be able to:
- Define critical control points (CCPs) with QC tests and check for products and processes
- Provide proactive alerts for QC tests in the warning range to take action prior to failure
- Provide proactive alerts for QC tests outside of pass/fail limits
- Provide proactive alerts for QC checks that are past due or have not occurred
- Hold shipments until the product has passed all QC and CCP tests
- Provide alerts to create lot retests after a specified age
- Use shelf-life and expiry dates to prevent the use or distribution of expired product.
“A comprehensive business management solution gives a manufacturer more control and accelerates its supply chain,” notes Jean Huy, VP, global product marketing at Sage Business Management Solutions, the provider of Sage X3 software. “In particular, the [software] system integrates quality controls at every step of procurement, manufacturing and inventory management and provides extensive traceability of materials and ingredients to help reduce the risk of a recall or the scope of a recall, if one is necessary [tracking back to the exact ingredient, lot and sublot that caused it].”
Undoubtedly, quality management plays a big role in managing recalls. A quality management solution can be provided as a standalone system or as a module within an ERP system, according to Mike Edgett, Infor industry & solution strategy director, process manufacturing. But, there are other tools that can help prevent recalls. “A product lifecycle management [PLM] solution, such as the Infor Optiva solution, not only helps in the product development cycle, it also ensures the label or specification sheet shipped with a product exactly matches what’s inside the package,” continues Edgett.
Shop floor control
What you once may have thought of as a food and beverage shop floor system (e.g., batch control and inventory management) today offers much more control of food safety and quality management. Plus, it includes tools that can prevent unsafe product from exiting your premises and monitor a recall, if necessary. For example, the CAT Squared system uses real-time serialized management of all received inventories—including raw spices, dry goods and meat (both live animals and raw meat)—to track all movements within a facility, says Brandon Murphy, CAT Squared professional services manager. “Our scaling and batch-mixing applications serialize all cases and pallets, creating a direct link from the final client label and your internal lot number to the originating vendor lot for each item [or ingredient] included in your final product.”
With the CAT Squared system, processors know where all their inventory is at all times, as well as what items are being mixed together, what is being produced, what is being stored and where, and what is being shipped. The system can be expanded with a real-time food safety and quality management (FSQM) system and real-time laboratory information management system (LIMS). By adding these systems, processors can make products on positive release holds, preventing the shipment of serialized inventory until all preapproved lab tests and quality checks have been performed.
The InfinityQS enterprise quality management platform can be used for recording key quality data, HACCP checks, microbiological tests and labeling verifications across a wide variety of food and beverage applications, according to Steven Voight, client solution engineering manager. “On the plant floor, our solution provides real-time data monitoring and alerts, allowing operators to identify and resolve quality issues and violations before they become larger problems.” In addition, processors can “roll up” and analyze the data within the platform’s centralized repository and identify areas for improvement that will proactively drive the production of highquality goods and prevent costly recalls.
Enterprise manufacturing intelligence (EMI) systems, such as the Rockwell Automation FactoryTalk solution, pull control-level data and collect and contextualize it in meaningful, role-based dashboards. This gives an array of stakeholders access to dive into what’s actually happening within a facility, explains Dan Updyke, Rockwell Automation batch product manager.
“EMI solutions are enhanced when connected with an MES,” notes Updyke. “Operators are required to conduct the correct test based on verified product and process-order information, and collect and archive test results for each production run. The software can integrate quality management into an operator’s workflow, easing regulatory compliance and enforcing operational standards.”
Predict risk, prevent a recall
If a processor has an HACCP/HARPC plan in place, physical steps can be taken to reduce risk and prevent recalls. For example, in small plants where separate coolers aren’t available, raw meats must kept on separate pallets on the floor, with packaged RTE foods above them, says Scioscia. “Relying on the integrity of packaging as the only precaution against cross-contamination is not a wise decision.” In addition, pickers need to be trained to identify packaging issues as well as allergens and how to store allergens so they don’t contaminate other food products.
The primary tool to prevent risk initiates within the quality program, says Joey Benadretti, president of SYSPRO USA. Inspections of ingredients and products that identify deviations outside acceptable levels should trigger alerts to address these problems.
Any risk assessment should identify weak links and define the CCPs, states Payne. The CCPs can be monitored with proactive alerts and analytics to determine whether tests in certain areas of receiving, inventory, manufacturing and the shipping processes are registering high failure rates.
“[Our] Business Activity Monitoring module can be configured to monitor for variations in production or anomalies at quality checks. It also can alert people or groups when those anomalies are found,” says Griffin. When linked with an ERP system, a warehouse management system (WMS) can ensure proper labeling is being used and prevent users from generating labels incorrectly.
“All the members of our implementation staff take the same HACCP for Processors training that our clients do,” says CAT Squared’s Murphy. This allows the staff to understand the process and approach clients have taken to develop their HACCP plans. “During the implementation process, we analyze all data collection and analysis methods being used and either replicate them into a digital real-time format or work with the plant to adjust its plan to best take advantage of the tools we provide,” adds Murphy. Monitoring CCPs in real time through machine integration and SPC trending of data and alarm limits (separate from critical failure limits) lets a manufacturer know if a process is trending out of control before a critical failure occurs.
At the ERP level, the integrated QC control capabilities of Sage X3 software ensure the control of raw material, finished goods and in-process manufacturing. With the software, inprocess procedures, notes and instructions can be documented on recipes/ BOMs and routings. Other procedures and documents concerning CCPs and corrective actions can be accessed from the collaborative capabilities of the ERP system, explains Huy.
Recalls: Keep them small
Many of the tools that can prevent a recall also can manage one. “[For instance,] SYSPRO’s ability to trace ingredients from origin through the manufacturing process to the ultimate customer helps companies fully comply with FDA rules and regulations pertaining to food safety,” says Benadretti. Using customer relationship management (CRM), with its combination of alerts, messaging, emails and possibly phone calls, allows the customer to immediately remove tainted product from shelves, dispose of it or return it to the manufacturing facility.
“A multitude of products can track finished goods once they’ve been shipped,” says Scioscia. For example, GPS devices can track the location and the temperature of a truck. In addition, when coupled with fleet management software, these GPS units can help drivers plan the most efficient routes for delivery. Proof-of-delivery applications can also verify a delivery was made. And, if there is a recall, this same data can be used to locate all the defective products that were delivered.
“The big thing is identifying where the product went,” says Deakins. “It may not be a single destination.” For instance, if a batch of products was placed on multiple pallets, they most likely went off in different directions. After a lot number is keyed into the ERP system, every place the particular lot number went to should be revealed, and the system can immediately notify all the affected people in the supply chain, says Deakins. Processors that want to save valuable time may ship products “at risk” (meaning they are technically on hold by the QC department until all test results are in). Prior to products being shipped at risk, the customers and processor must enter into an agreement that the customers will not place them into circulation until they receive an OK from the processor’s QC department indicating the products have passed all tests and are off hold, according to Deakins.
With lot traceability, a processor can identif y the lots involved in a recall, notify all members of the supply chain to stop moving a defective product and contact retailers to pull it off the shelves. Some retailers, like Costco and Wegmans, can use their loyalty card programs to contact customers who may have purchased a bad product.
Sometimes, a processor may need professional help to manage a recall. Companies like Stericycle ExpertSOLUTIONS offer a comprehensive suite of services to help processors manage recalls throughout the entire recall lifecycle, including preparation, notification, remedy management and disposal, according to Kevin Pollack, Stericycle vice president. “We assist with identifying and notifying affected parties throughout the supply chain. Once an appropriate remedy, such as a refund or coupon, is identified, our team can manage its distribution. Our scalable contact center can be quickly ramped up to handle an influx of calls that might overwhelm an internal call center. We also remove affected product and conduct regular effectiveness checks to monitor the process for accuracy to help ensure compliance with FDA and USDA. Finally, our state-of-theart infrastructure can manage and store any returned product until closeout with regulatory bodies is completed and offer innovative, sustainable solutions for the disposal of affected product.”
Keep in mind the better connected your software tools are, the easier it will be for you to prevent a recall. And, if you need to start a recall, these same tools can make it a much smoother process.
For more information:
Joe Scioscia, VAI, 800-824-7776,
jds@vai.net, www.vai.net
Jay Deakins, Deacom, 877-433-2266,
info@deacom.com, www.deacom.com
Jack Payne, Aptean, 770-351-9600,
jack.payne@aptean.com, www.aptean.com
Brandon Murphy, CAT Squared, 501-328-9178,
brandon.murphy@catsquared.com, www.catsquared.com
Dan Griffin, Exact Macola, 800-468-0834,
info@exactmacola.com, www.exactmacola.com
Jean Huy, Sage Business Management Solutions, 604-207-3611,
jean.huy@sage.com, www.sage.com
Mike Edgett, Infor, 800-260-2640,
mike.edgett@infor.com, www.infor.com
Dan Updyke, Rockwell Automation, 440-646-3434,
daupdyke@ra.rockwell.com, www.rockwellautomation.com
Joey Benadretti, SYSPRO USA, 714-437-1000,
info@us.syspro.com, www.syspro.com
Kevin Pollack, Stericycle ExpertSOLUTIONS, 888-732-3901,
info@stericycleexpertsolutions.com, www.stericycleexpertsolutions.com
Speeding up QA data
When quality assurance lags behind production, the entire process is held up. That’s why it is important to minimize the inefficiencies created by manual data reporting. In other words, operators and quality assurance teams need real-time trends and reports.
For instance, a Hillshire Brands plant in Texas that produces more than 50 varieties of meals on a stick required numerous changeovers involving modifications to equipment. Quality deviations and wasted product were occurring due to the process variables not being properly changed for each new recipe and downstream areas of production that were not prepared for the increased or reduced time frame that came with the changeovers. Since the plant depended on manual data entry, any deviations were not discovered until after a shift had concluded. Then, quality assurance managers had to sort through the data to find out where and when the issue arose, and how much product of which variety needed to be thrown out.
Now, the plant has a FactoryTalk EMI solution in place, which displays real-time information via dashboards. Operators and value stream managers on the floor can monitor any area of production and even share relevant information in real time. And, if there’s a concern about food safety, quality assurance can immediately pull up data to determine how many cases have been affected.
Source: Rockwell Automation.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!






