Food Safety
Automated microbiological testing helps speed up determining if food is safe
The adoption of automated or rapid methods for pathogen testing should include some kind of overall assessment of potential benefits and risks.

When it comes to pathogen testing, processors must choose from a number of processes, as well as whether the work will be done in-house or through a contract laboratory.
Photo courtesy of Getty Images/Luchschen

Testing samples of raw or finished products for pathogens presents processors with a number of challenges, including the need to ensure that contract laboratories have equipment properly designed and calibrated to do the specific tests needed.
Photo courtesy of Getty Images/AlexRaths
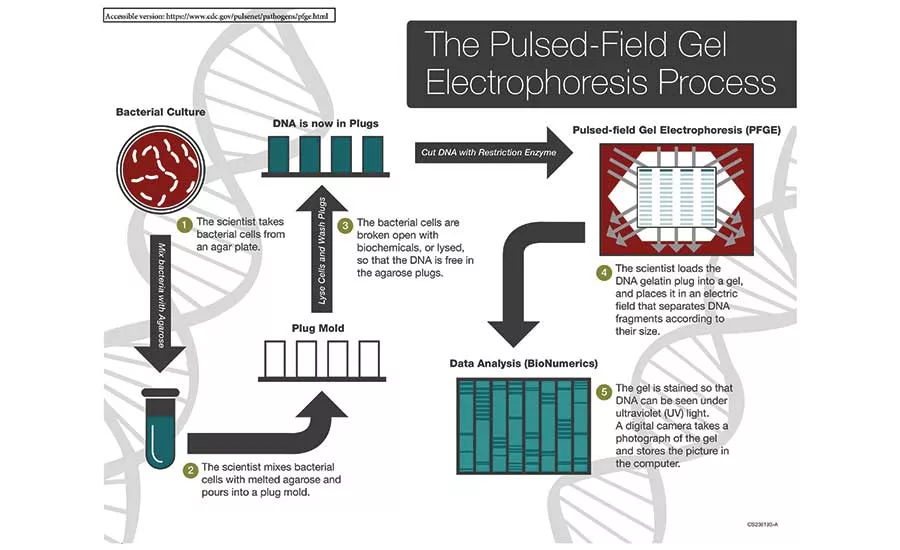
A diagram of the Pulsed-Field Gel Electrophoresis process.
Source: Centers for Disease Control and Prevention
There is an old saying that “all things must pass.” One of the things that unfortunately seems to have passed is the rapid methods workshop.
For 30 years, ending in 2010, Dr. Purnendu Vasavada organized and hosted a rapid methods workshop at the University of Wisconsin, River Falls. Dr. Dan Fong conducted a similar workshop at Kansas State. These programs allowed people in the field and other interested parties to remain up-to-date as to what was old, new and on the horizon in microbiology and its applications in the food industry. It was truly one-stop shopping, augmented by demonstrations of the different technologies by the vendors of these different technologies. So, one must dig a little more today to find that information.
Automation in food microbiology
Given the nature of food, a fully automated tool for microbiological testing and analysis is not something that will be seen today or in the near future. Many of the things seen in the old Star Trek episodes have come to pass, but the bio-filter which identified and removed specific pathogens and other contaminants as people and things were “beamed up” is not one of them. Yes, there are biosensors that can detect certain organisms or metabolites, but their applications to foods and processing systems are limited. Before food samples can be tested, they have to be prepared in some way, shape or form. Automating this step will always be a challenge, but once samples have been prepared, there are currently many different automation options. These include plating, reading plates, inoculation of different media, reading cultures and monitoring growth.
However, do keep in mind that when considering the adoption of methods in your own shop or looking for a contract laboratory to do work for you, it is best to use—or ensure that the contractor use—official or recognized microbiological methods. Today, the expectation is the contract laboratory will be ISO 17025 certified. A great source for testing information is the United States Food and Drug Administration’s BAM Manual, or Bacteriological Analytical Manual. (Find the link to the BAM Manual at the end of this story.)
As with all food quality and safety operations, the adoption of automated or rapid methods should include some kind of overall assessment of potential benefits and risks. Automation by definition implies that tasks may be done more quickly and easily with less human intervention. This potentially will allow a company or laboratory to make better use of their human resources. There is also a potential for reducing human error assuming that the equipment used for automation is properly maintained and calibrated. Let’s look at some of these applications and the available systems.
Sample Preparation: As noted above, sample preparation is always a challenge, especially when working with solid foods. The samples must be collected, handled aseptically, weighed and diluted and mixed. One of the ways of speeding this up includes the use of prepared diluents; that is, diluents that have been weighed and measured and simply require the addition of the sample. Other means of automation include the use of automatic dispensers for adding the diluents (automated pipetting). Automated dispensers can save time and effort, but they must be calibrated regularly and records of calibration maintained.
Media Preparation: Preparation of media in advance or as needed is another means for automating operations. Laboratories can purchase a wide range of different media, but there is a small chance that such media could be contaminated in transit, storage or handling in the laboratory. There are also many different systems for preparing, sterilizing and dispensing media. These include more sophisticated systems that may be programed to handle sensitive components.
Dispensing and Plating of Media: Systems for media preparation may also be linked to dispensing and plating systems that will inoculate plates, dispense the appropriate media at the proper temperature and ensure the inoculum is evenly distributed. These systems also may be set up to label and stack plates.
It is imperative that processors or laboratories that decide to automate be sure that the system they adopt and the media that they are using has been validated for the food system or product that they are testing.
As an example, a manufacturer of different grains, including flax seed, began working with a new contract laboratory and suddenly found that their products were out-of-spec for aerobic plate counts. They submitted duplicate samples to other laboratories and all were within specification. It turned out that the contract laboratory had recently begun using an automatic plating and reader system, which was giving artificially inflated counts. Apparently, the starch granules in the seeds were swelling and being read as colonies. And, the manufacturer had suggested that the system not be used for grains.
Automated dispensers and platers have been used for many years. One of the old stand-byes is the Spiral Plater, but there are now other more sophisticated systems available to the industry.
Plate Reading: When reading plates is done manually, it is tedious and requires proper training. Reading plates lends itself perfectly to automation, provided the systems are properly utilized. Otherwise, you get an issue such as the previously mentioned starch granules.
Automated systems today allow much greater flexibility in that they can be set up with different lighting, allowing the user to count only colonies that exhibit certain characteristics such as fluorescence. Readers can also be linked to cameras and computers. This allows counting to be recorded automatically, but also allows plates to be examined under higher magnification to better enumerate what is there.
Monitoring Turbidity: Growth rates in liquid media may be monitored by changes in turbidity. The bottles used in these studies are specific for the individual systems and what is being enumerated or sought. These are utilized more for medical work than food. Suppliers of such systems include BacT/Alert from BioMerieux and BACTEC FX.
The bottles are inoculated and placed in units where temperatures are controlled and the bottles continuously monitored. Cliff Coles, Clifford Coles Food Safety Consulting, Inc., says that the advantage of the BacT/Alert is that the sample is under active incubation, whereby the bottles are gently rocked in the system. This provides more oxygen that bathes the recovering, potentially sub-lethally injured cells with a continuous supply of nutrients.
Other systems use a passive incubation where the samples are idle during incubation. The Bactometer system utilizes impedance measurements, or the change in electrical current caused by microbial growth and metabolites. Introduced in 1970, the work done by Dr. Ruth Eden on impedance was ground breaking. The system did require a standard curve to be done for each product to establish a minimum time to detection but still it was a great advance for food microbiology.
Colorimetric Changes: The same principles and set apply if one wants to monitor colorimetric changes. BacT/Alert monitors colorimetric changes and reflected light to monitor carbon dioxide in the samples which is related to growth of microorganisms. The production of carbon dioxide causes a gas permeable sensor to change color.
The BacT/Alert system is commonly used in the food industry to monitor the spoilage potential of many aseptic products. Companies will utilize the system and hold product until the results indicate that spoilage will not be an issue. If there are positive samples, the bottles may be recovered and cultured.
The BacT/Alert was introduced to the food industry by Coles when he was running California Microbiological Consulting Inc., so he has unique insights into the system. Coles gives the example of Del Monte Corporation needing a rapid, reliable system to determine if aseptic products such as tomato paste, pudding cups and other products were contaminated. Studies conducted on tomato paste indicated that the BacT/Alert could detect down to a few cells within 8 to 10 hours. This not only detected compromised samples rapidly, but also greatly reduced the response time to mitigate a compromised aseptic system.
Prior to BacT/Alert it was an industry standard to hold aseptic product in a warehouse for up to 10 days in order to determine if swells would develop. Obviously, the cost of holding product and the real estate needed to hold product was a concern; not to mention that if there was spoilage, then everything produced 10 days previously and produced up to that point was most likely going to spoil. The Bac-T/Alert system has become a standard for detecting spoilage in the tomato paste industry as well as for other aseptic commodities.
The United States Food and Drug Administration does not require that firms producing low-acid aseptic products conduct incubation studies. However, if an investigator is in a firm producing aseptic products and the company is doing incubation studies, they are supposed to do the following:
- Determine whether the containers are statistically sampled;
- How many containers are incubated;
- Time and temperature of incubation;
- Firm specifications for acceptability of lot;
- If spoilage is detected by firms, do they perform spoilage diagnosis to determine cause;
- Specify method and describe lot disposition procedures.
There is a reason that the Agency does not require incubation studies, and it’s the same reason that the HACCP (Hazard Analysis, Critical Control Points) concept was created so many years ago and has been mandated not only in the United States, but the world over—one cannot test safety into a product. Safety must be built into the processing system and that system properly monitored and maintained. To quote the National Academy of Sciences 1985 report entitled “An Evaluation of the Role of Microbiological Criteria for Foods and Ingredients:”
“HACCP provides a more specific and critical approach to the control of microbiological hazards than that achievable by traditional inspection and quality control procedures.”
In other words, finished product testing does not properly ensure safety, but it may be used to verify that a system is working as designed.
Pathogen testing
There is a greater emphasis today on pathogen testing in the food industry. This is due in large part to the Preventive Controls Regulation (21 CFR Part 117) that was enacted in the Food Safety Modernization Act, especially the clause that mandates environmental monitoring for some products and processes. The Preventive Controls regulation in 21 CFR Part 117.130 (c) (2) states the following:
“(ii) The hazard evaluation required by paragraph (c)(1)(i) of this section must include an evaluation of environmental pathogens whenever a ready-to-eat food is exposed to the environment prior to packaging and the packaged food does not receive a treatment or otherwise include a control measure (such as a formulation lethal to the pathogen) that would significantly minimize the pathogen.”
So, the bottom line is that there are many operations that must now do environmental monitoring for Listeria or Salmonella depending upon the product. Most companies will decide not to do this work in-house, so they utilize a contract laboratory to help them set up the program and do the actual testing. Contract laboratories have the sample volume and resources to do the work, which lends itself to utilizing one or more automated systems and/or adopting rapid test systems aimed at screening for potential pathogens.
Environmental monitoring is not the only area where pathogen testing is conducted. More and more companies are demanding that their vendors provide them with Certificates of Analysis (COA) that include pathogen testing, especially salmonella, Listeria monocytogenes and pathogenic E. coli strains.
“It is imperative that the laboratory be ISO 17025 certified to conduct the tests that are being requested and that you are informed of the method(s) being used,” says Coles. “In a number of cases initial false positive results have led to unnecessary product recalls and market withdrawals. Confirmatory testing is as critical to a diagnosis as is the initial suspect positive test.”
Automated tools for pathogen testing and identification include enzyme immunoassays (EIA), PCR (Polymerase Chain Reaction) testing, Pulse Field Gel Electrophoresis (PFGE) and several new technologies which are being evaluated in the clinical arena. The BAM Manual includes PCR test methods for Molecular Detection of Cyclospora cayetanensis in Fresh Produce (Method 19B) and Detection and Quantitation of Hepatitis A Virus (Method 26A).
To put it simply, enzyme immunoassays or enzyme linked immunosorbent assays (ELISA) are tests that utilize an enzyme that is attached to a substrate to detect an antigen in a sample. The antigens bind to the enzyme, a specific binding agent (another antibody) is added and, lastly, the enzyme’s substrate is added. This produces a detectable color or light reaction which is read by a sensor. These tests can be used for microbiological testing, but also form the basis of the allergen testing so prevalent throughout the food industry today. There are currently three types of ELISA tests—direct, sandwich and competitive.
Polymerase chain reaction (PCR) is a system that allows a single chain or segment of DNA to be multiplied thousands to millions of times. The technology generally utilizes a heat stable DNA polymerase, which assembles the DNA chain from free nucleotides. The technology has many applications, including medical, infectious diseases and forensics. The forensics applications vary from examining ancient peoples to identification of foodborne pathogens.
Pulse Field Gel Electrophoresis is included in this discussion since it is one of the primary tools used by PulseNet along with Whole Genome Sequencing (WGS) and Multiple Locus Variable-number Tandem Repeat Analysis (MLVA) to identify isolates from foodborne outbreaks. Once the pathogen has been identified and sequenced, it goes into the PulseNet database. If like strains pop up in other locations, whether they are from food, a production operation or from sick persons, the PulseNet system flags the match to indicate an outbreak. At the moment, PFGE is considered the gold standard for fingerprinting, but CDC has indicated that they are transitioning to Whole Genome Sequencing. PFGE works as follows (GDC, 2018):
- The scientist takes bacterial cells from an agar plate;
- The scientist mixes bacterial cells with melted agarose and pours into a plug mold;
- The bacterial cells are broken open with biochemicals, or lysed, so that the DNA is free in the agarose plugs;
- The scientist loads the DNA gelatin plug into a gel, and places it in an electric -field that separates DNA fragments according to their size;
- The gel is stained, so that DNA can be seen under ultraviolet (UV) light. A digital camera takes a photograph of the gel and stores the picture in the computer.
Once the DNA fragments (fingerprints) are created and stored, they are analyzed by a program called Bionumerics. Once the analysis is completed, the fingerprint is uploaded to the national database where it is accessible to current or future epidemiological investigations. The PulseNet database, which was established in 1996, has been instrumental in initiating investigations that may well have stumped scientists before the system was brought on-line. Perhaps the most interesting and one of the earliest applications of the system occurred with the outbreak attributed to cereals more than 20 years ago. There are currently 83 laboratories across the United States that make up PulseNet USA with at least one facility in each state of the union.
Automation, food quality and safety
How individual food processors manage the microbiological element of their food safety, quality and sanitation programs depends upon each individual processor. Some may elect to utilize contract laboratories exclusively, others may decide to work in concert with the contract laboratory by collecting samples or doing some preliminary work and others may decide that they want to conduct all or part of the microbiological work in-house. The “part” element of the equation usually occurs because a processor simply does not want to deal with pathogens in-house, so operators may do some preliminary work, yet send presumptive positives to an outside laboratory for confirmation. Making the decision to work with pathogens in-house changes the whole dynamic of the microbiological laboratory.
As noted, it is usually only large operations or corporations that decide to do microbiology in-house. They may elect to automate, as does almost every contract laboratory. The latter do so because of volume and a desire to reduce costs. Processors that decide to utilize outside laboratories should select their contract laboratory in the same way that they evaluate and select any one of their suppliers. The following is a list of potential criteria that a processor should consider when it comes to selecting and evaluating a laboratory partner (Stier, 2016). There may be more or there may be fewer criteria. That will depend upon the processor and their needs.
- The laboratory has the ability to do the work needed using official and recognized methods.
- The laboratory is ISO 17025 certified.
- The work can be started as soon as the samples arrive and results will be available as the testing protocol dictates.
- The laboratory can provide references that the company may speak to.
- The laboratory will pick up samples.
- The laboratory allows audits.
- The company has trained employees that participate in check sample programs.
- The laboratory has experience with your product matrix or matrices that are very similar.
- The laboratory will not only provide results, but will interpret results if needed.
- The laboratory demonstrates a sense of urgency with your samples.
- The laboratory has the ability to suggest possibly more appropriate tests in addition to or in place of the testing you are asking for.
- Cost of services.
An integral part of this process should be a visit to the facility where the company can see exactly what they do. Are they using automated techniques? Have the methods been validated against the matrix you are manufacturing? Will they work with you and help you do what is necessary? Does the lab have the ability to diagnose what may be causing the problem or do you only get a sheet of results and you are on your own to decipher what the microbial tests are actually telling you? (Keep in mind that one key reason you use an outside lab is to get assistance in solving problems, not just to get data.) Will they work with you and help you do what is necessary? This last point is extremely important for the small processor as they usually do not have the resources of large companies.
When looking at utilizing automated methods, whether a laboratory or food processor, it is imperative that these methods be properly evaluated to determine their value to the company and validated to ensure their accuracy. The BacT/Alert is an excellent tool for evaluating sterility with aseptic systems, but it is a tool for verifying the process. The keys to producing safe and wholesome aseptic products are proper monitoring and maintenance of processing systems, sterilization of packaging systems and sterilization of the filling areas, plus ensuring that the packages are properly sealed. Tests like PCR and EIA are forensic tools to identify for the microbiologist the pathogen or spoilage organism.
But even with all this technology and sophistication, the food industry still needs food microbiologists who are learned in the basics; what bacteria are associated with what foods, how these organisms can be controlled in the field or pasture, in transit and in the plant, and how we can manage them to ensure a safe food supply with a minimum of losses that can be blamed on the “bugs.” There is still a place in the industry for someone who is not a gene jockey.
Click here to read our interview with LexaGene CEO Dr. Jack Regan >>
For more information:
USFDA, (2018), Bacteriological Analytical Manual (BAM)
https://www.fda.gov/Food/FoodScienceResearch/LaboratoryMethods/ucm2006949.htm
USFDA (2014), Aseptic Processing and Packaging for the Food Industry
https://www.fda.gov/ICECI/Inspections/InspectionGuides/ucm074946.htm
USFDA, (2015), Rapid Methods for Detecting Foodborne Pathogens (Archive Method)
https://wayback.archive-it.org/7993/20170406182241/https://www.fda.gov/Food/FoodScienceResearch/LaboratoryMethods/ucm109652.htm
Centers for Disease Control and Prevention, (2018), Pulsed-Field Gel Electrophoresis
https://www.cdc.gov/pulsenet/pathogens/pfge.html
Dr. Purnendu Vasavada,
purnendu.c.vasavada@uwrf.edu
Clifford Coles, Clifford Coles Food Safety Consulting Inc.
cmlkcoles@gmail.com
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!




